One of the most important features of polyacrylamide is its flocculation property in the treatment of various industrial effluents at high densities and high molecular weight. Therefore, its main application is in the water and wastewater treatment industry. The use of polyacrylamide in this industry reduces the use of other conventional coagulants, which results in a much lower amount of sludge and makes the process highly economical and cost-effective.
Use of polyacrylamide in the water and wastewater treatment industry:
The drinking water we use every day is obtained from the treatment of natural water. In the treatment process, polyacrylamide is generally used to remove bacteria, viruses, and algae, and also in the hydrolysis process, anionic polyacrylamide is used. Used in low-grade water purifiers. Since the presence of sodium in raw water is mainly due to colloidal particles, dissolution and filtration are required, so low molecular weight flocculants are used.

The use of this polymer can reduce the precipitation action and control the acidity. In general, the most important applications of this polymer can be summarized as follows:
- Role in helping coagulation such as the process of clarifying and purifying.
- Coagulation of solids and organic compounds in the return water of heating systems
- The role of coagulants in industrial and sanitary wastewater treatment systems.
Polyacrylamide in the role of flocculation agent in wastewater treatment industry:
In industry, the method used to remove suspended impurities and colloidal substances in water and wastewater is mainly coagulation and flocculation. To do this, we need a coagulant that neutralizes the charge of the colloidal particles, bringing them closer together and forming heavier particles in relatively larger dimensions, polyelectrolytes, or more precisely polyacrylics. Amide can do this. Suspended and colloidal particles are large enough and can be easily precipitated and smoothed. Purification is performed by this mechanism and the key role of this polymer in it is clearly understood.
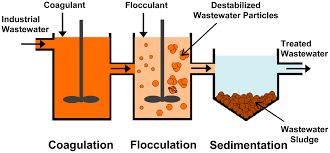
This polymer has a moderate moisture absorption property and if it is not exposed to high temperatures, its powder can be stored for a long time. In solution, when its concentration is more than 17% it can be stored for more than a year without significant change in viscosity.
Read more: Caustic Soda in water treatment
Synthesis of anionic polyacrylamide:
Anionic polyacrylamide can be prepared by the anionic polymerization method. The polymerization process of acrylamide monomer is done by using a kind of radical initiator and sodium hydroxide as a hydrolysis agent.
To be more precise, for the synthesis of the samples, a solution of acrylamide monomer is first prepared in water and then a certain amount of sodium hydroxide is added to it and the reactor temperature is raised to 35 ° C, the reaction continues for 30 minutes. To get rid of the oxygen, we add the initiator in the next step, and it is better to use isobutyro nitrile for this reaction.
Then increase the reactor temperature to 80 ° C and give it 4 hours to complete the reaction, in the last step, specialization and drying operations are performed to produce the product in the form of a uniform powder.
How to dissolve polyacrylamide in water?
The solution of dissolving solid polyacrylamide in water can be either a simple task or can be difficult or complex depending on the situation. Polyacrylamides are particles with very-long-chain lengths, very high molecular weights, and are very hydrophilic. For easier solubility in water, these particles are prepared and supplied in the form of granules and powder.
When dissolving polyacrylamide in water, pay attention to the following points:
The rate and rate of dissolution of polyacrylamide in water depend on several factors, such as the temperature of the water, the speed of the stirrer, or the molecular weight of the polyacrylamide and their electrical charge.
powder particles of this polymer dissolve easily in water but will need more time to dissolve if these particles stick together due to moisture.





