Description
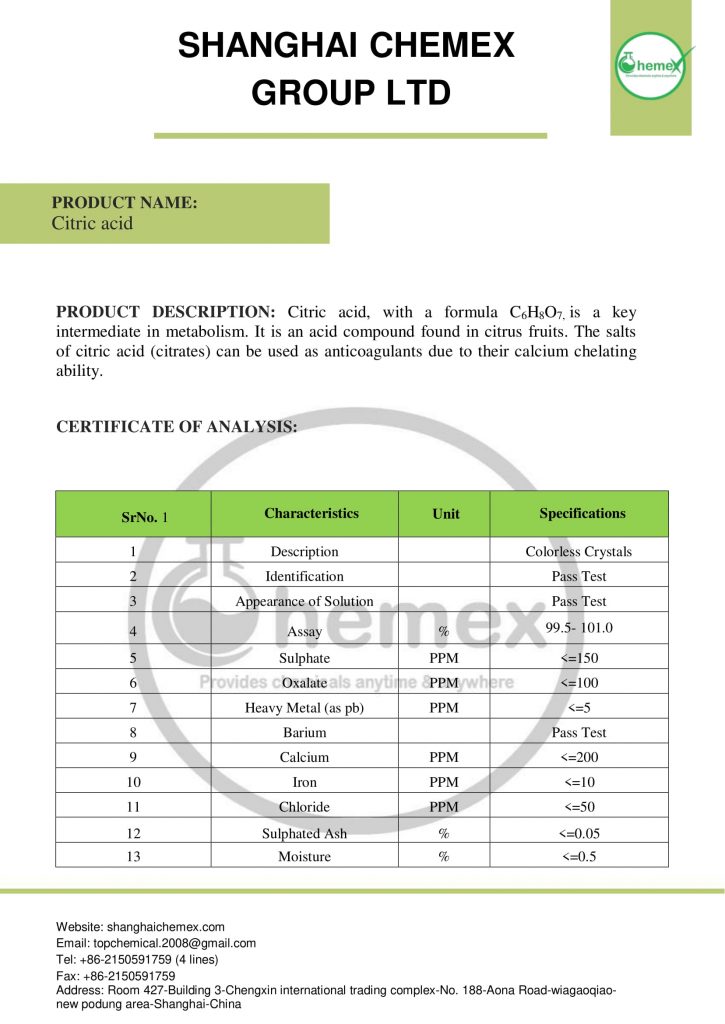
Citric acid is a weak organic acid with the chemical formula C6H8O7, which is available in both industrial and food grades and is found in many foods such as fruits and vegetables. Pharmaceutical, health, oil, gas, detergent, and food products all contain different amounts of citric acid. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties of Citric Acid:
The appearance of this compound is a solid, crystalline, and white powder with a melting point of 153 ° C. This acid is divided into two categories: dry citric acid (anhydrous) and aqueous citric acid (monohydrate). The following table lists some of the important chemical and physical properties of this compound:
| Chemical formula | C6H8O7 |
| Molecular Weight(g/mol) | 192.123 (anhydrous),
210.14 (monohydrate) |
| Appearance | solid |
| odor | Odorless |
| taste | Strongly acidic taste |
| Density (g/cm3 at 25 °C) | 1.665 (anhydrous),
1.542 (monohydrate) |
| Melting point (° C) | 153 |
| Boiling point (° C) | 310 |
| color | white or colorless |
| form | crystals or powder |
| Chemical Structure Depiction | 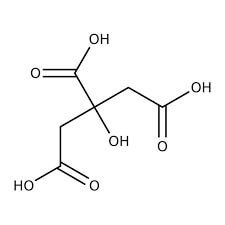 |
The Key Difference between Anhydrous and Monohydrate Citric Acid?
This compound is available in two forms, citric acid monohydrate and citric acid anhydrous, The difference between these two acids is one in their granulation, whose monohydrate, crystalline, and anhydrous is a powder, another difference in their processing is that monohydrate is formed from the crystallization of cold water and that anhydrous is formed from the crystallization of hot water.
Due to the structure and properties of this compound, almost different uses have been defined for them. For example, its monohydrate can be used in a variety of sour-tasting beverages, and its anhydrous one can be used as an acidic pH regulator, creating a sour taste and improving the glossy coating of the fruit surface.
Preparation of Citric Acid:
One way to prepare citric acid is to extract it from citrus and sour fruits. But as mentioned, this method has no economic and time justification and is not used on a macro level. How to prepare citric acid is done in two main ways around the world, which are explained below:
-In this method, a fungus called Aspergillus Niger is used. These fungi are placed in specific culture media at a temperature of 30 ° C. In these special conditions, Niger begins to produce citric acid by fermenting the substance in its bed, and after a few days, the entire surface of the bed will be filled with citric acid.
-One of the methods that can be used to achieve citric acid is the use of chemical reactions that use glycerin. This type of reaction is less exploited due to the time-consuming and low volume of the final product compared to the fermentation method using Niger fungus.
Citric Acid Uses:
- Due to its acidic and tasteful properties, this compound is mainly used as a flavoring and preservative, especially in beverages and sweets.
- Used to stabilize or maintain drugs and as a disinfectant against viruses and bacteria.
- Citric acid can be used as an alternative to nitric acid in deactivating stainless steel.
- It can also be used to purify water, which improves the effectiveness of soaps and detergents. Citric acid chelates metals in hard water to allow detergents to foam and work better without having to soften the water.
- Due to the presence of citric acid, you can adjust the pH level of your crop. You will also be able to check the quality of the flavor of fruits and vegetables.
- It acts as a natural scrub and is very effective in removing skin pigments. This acid is an antioxidant that reduces the aging process.
What is the Role of Citric Acid in Food Additives?
Citric acid is used as a food additive in factories; Currently, the demand for this solid acid is high, and for this reason, the market for buying and selling it in food-grade is booming. Production of jellies, jams, sweetened beverages, edible oils, frozen foods, canned goods, meat products, sweets and chocolates, dairy products, etc. are products that use some kind of citric acid.
This acid also causes a sour taste and a pleasant taste in Fruit leather, pickles, and soft drinks; It is also responsible for regulating the acidity of the food environment and eliminating the activity of harmful bacteria. Like Triammonium citrate
The Citric Acid In Pharmaceutical and Cosmetics:
Citric acid is used in the formulation of some cosmetic products. The main role of this acid is to regulate the acidity of the reaction medium; For example, acid is used to create a neutral environment in anti-dandruff shampoos and to brighten hair. Another application of citric acid is its use in the production of various drugs such as effervescent tablets, aspirin, and medicinal condiments.
Global Market:
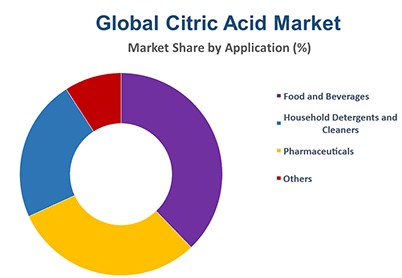
Citric acid due to high global demand is one of the largest fermentation products that is produced in the world with many tonnages. about 75% of the citric acid produced in the world is used in the food industry, and other amounts produced are used in pharmacy and industry.
The largest share of citric acid production belongs to European countries. After Europe, the largest producers in the world are the United States and China. Given the low price of citric acid supplied to the world market by China, it can be said that China has been able to compete very successfully with other producers of this acid, and by eliminating some competitors, a significant share in this field to Allocate yourself. Shanghai Chemex in China is one of the most reputable manufacturers of this chemical in the world.
Safety Information:
Given that this acid is used orally in many food products, it does not pose much of a risk to the gastrointestinal tract, but it is important to know that eating large amounts of Citric acid causes vomiting and heartburn. In this case, it is better to drink about 300 ml of water and then milk and see a doctor if necessary. Due to its properties, this acid can irritate the eyes and even the skin in high concentrations and cause chemical burns and blisters on the skin surface. In case of eye or skin contact with this acid, the contaminated surface should be washed immediately with lukewarm water for 20 to 30 minutes.
Packing and Storage:
Keep the container tightly closed in a dry and well-ventilated place.








lara –
Is citric acid the same as vinegar?
china chemicals –
While both are acids, they are not the same. Citric acid is an organic acid, while white vinegar is acetic acid.