Description
Disodium Phosphate is a chemical compound with the formula Na2HPO4.xH2O. It is a salt of disodium phosphoric acid that is used as a food additive, protein stabilizer, buffer, suspender, and accelerator. Disodium phosphate and trisodium phosphate are phosphate salts that are used to regulate the pH, absorb water in food products such as cheeses, condense milk, and in the production of pasta. The main industries consuming this chemical include detergent factories, water purification industries, food industries, ceramics industries, mining industries, paper production industries, and petrochemical industries. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
This product is a non-flammable compound that is easily soluble in water but does not dissolve in organic solutions. Disodium Phosphate can be produced in two forms: aqueous (12 water) in the form of white crystal and dry form (2 water) in the form of white powder.
The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | Na2HPO4 |
| Molecular Weight(g/mol) | 141.96 (anhydrous),
177.99 (dihydrate), 268.07 (heptahydrate) |
| Appearance | White crystalline solid |
| odor | odorless |
| Taste | Saline taste |
| PH (1 % solution) | Between 8,4 and 9,6 |
| Density (g/cm3) | 1.7 |
| Melting point (° C) | 250 (482 °F; 523 K) |
| Water Solubility | Freely soluble in water |
| Solubility | insoluble in alcohol |
| Other names | Disodium hydrogen phosphate,
Disodium hydrogen orthophosphate, Sodium phosphate dibasic |
| color | Colorless, translucent, or white |
| form | Crystals or powder |
| Chemical Structure Depiction | 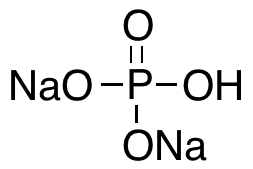 |
The Production Process of Disodium Phosphate:
- Disodium Phosphate is made by a simple chemical reaction between phosphoric acid (H3PO4) and sodium hydroxide. Phosphoric acid is a mineral acid. It is neutralized by sodium hydroxide, which acts as an alkaline substance during the reaction. Sodium atoms replace two atoms of the three hydrogens in phosphoric acid and disodium phosphate.
H3PO4 + 2 NaOH → HNa2PO4 + 2 H2O
- The compound is industrially produced in a two-step process using dicalcium phosphate with sodium bisulfate, which precipitates calcium sulfate.
CaHPO4 + NaHSO4 → NaH2PO4 + CaSO4
In the second step, the monosodium phosphate solution is partially neutralized:
NaH2PO4 + NaOH → HNa2PO4 + H2O
Disodium Phosphate Uses:
- To improve food properties such as nutritional value and improve cooking
- Decreased high blood calcium levels or increased low blood phosphate levels
- In some detergents and cleaners
- In the production of some glass glazed ceramics, and also in leather tanning, textile industries, printing
- In cosmetics and personal care products, sodium phosphate compounds are used in the formulation of bath products, colognes, cleansers, mouthwashes, hair conditioners, hair dyes, shampoos, and skincare products.

What is the use of Disodium Phosphate in Food?
Phosphate-containing compounds such as oral sodium phosphate are used to improve food properties such as nutritional value and improve cooking. This phosphate compound is used in packaged foods such as pasta. It is also used as an emulsifier in some cheeses, in addition to traces of this product can be found in meat products, jellies, dry milk, sauces, and so on. In fish and seafood, disodium hydrogen phosphate is sometimes used to retain moisture.
Buy Disodium Phosphate:
Disodium Phosphate is ready to be delivered to you, dear customers, in 25 kg bags. Shanghai Chemex is ready to receive the orders of our dear customers, provide the required product with the best price and quality, and send it as soon as possible by a reliable transportation system.
Safety Information:
The product may cause irritation to the eyes, skin, and respiratory system.

First-aid measures:
- Eye Contact: In case of contact with the eyes, immediately flush the eyes with plenty of water.
- Skin Contact: Remove this material from the skin with plenty of soap and water.
- Inhalation: Remove to fresh air immediately. If not breathing, give artificial respiration.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Keep away from incompatible materials. Keep in a dry, well-ventilated area in closed containers. Protect containers from physical damage.








Reviews
There are no reviews yet.