Rubber is an elastic material that exists naturally and synthetically. The natural type emanates from a tree that grows in certain areas, while the synthetic type is obtained through processes performed on chemicals. Due to its properties of elasticity, durability, and high hardness, rubber is the most important material in the automotive industry. More than 50% of the rubber produced worldwide is used to make car tires. Rubber is made of elastomers or elastic polymers. These polymers have a high molecular weight and can be returned to their original state after applying various stresses and pressures to them.
Natural Rubbers:
Natural rubber is an elastic substance obtained from the latex sap of trees, especially trees that belong to the family of Hevea and Ficus trees. Technically, if we want to define natural rubber, we must say that natural rubber is an elastomer or elastic hydrocarbon polymer. This material is one of the types of tires that are used in various processes to produce various rubber products. Natural rubber is also known by the common names of Indian rubber, elastic gum, and rubber. A wide range of rubber products are produced from natural rubber in the world; Types of tires, engine components, latex products used in medical work, etc., are examples of these products.
Synthetic Rubbers:
Synthetic rubber is a type of synthetic elastomer that is often derived from crude oil. More than two-thirds of the world’s rubber production capacity, which is about 15 billion kilograms, belongs to the production of synthetic rubber. Most of the demand for synthetic rubber, like natural rubber, is in the automotive industry, which produces a variety of rubber profiles, car doors and windows, belts, and floor coverings.
Some kinds of synthetic rubber:
- Neoprene rubber or chloroprene
- Silicone rubber or polysiloxane
- Nitrile rubber or NBR
- EPDM Rubber or Ethylene propylene Diene monomer rubber
- SBR Rubber or styrene-butadiene rubber
- Butyl Rubber or isobutylene isoprene
- Fluorosilicone Rubber or FVMQ
What are the Components of Rubber?
- Fillers
- Plasticizers
- Accelerator activators
- Inhibitors
- Antioxidants
- Vulcanization accelerators
Accelerator:
In the rubber industry and all related industries, accelerators refer to all compounds that accelerate the vulcanization process of rubber and also cause this process to occur at lower temperatures. Accelerators include many materials in this industry, but sulfur and nitrogen compounds such as benzothiazole derivatives are the most widely used. Accelerators are divided into two types, primary and secondary, according to the properties of the final product.
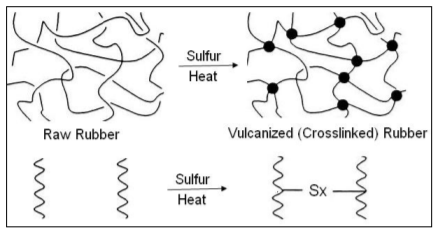
Vulcanization:
Vulcanization is the chemical process by which the physical properties of natural or synthetic rubber are improved. Finished rubber has higher tensile strength and resistance to swelling and abrasion and is more elastic in the temperature range. Triazole and sulfonamide accelerators are generally used as primary accelerators due to their unique properties. Sometimes secondary accelerators must be used to activate the primary accelerators. Some of the secondary accelerators are guanidine and dithiocarbamate. The rate of use of secondary accelerators is between 10 and 40% of primary accelerators.
Classification of Accelerators:
Each accelerator has its specific characteristics which help manufacturers to choose the best one for their final application.
| Accelerators | Chemical Group | Vulcanization Speed |
| BA, HMT | Aldehyde Amine | Slow |
| DPG, DOTG | Guanidine | Slow |
| MBT, MBTS, ZMBT Semi | Thiazole | Ultra-fast |
| ZBDP | Thiophosphate | Ultra-fast |
| CBS, TBBS, MBS, DCBS | Sulfenamides | Fast-Delayed action |
| ETU, DPTU, DBTU | Thiourea | Ultra-fast |
| TMTM, TMTD, DPTT, TBzTD | Thiuram | Ultra-fast |
| ZDMC, ZDEC, ZDBC, ZBEC | Dithiocarbamate | Ultra-fast |
| ZIX | Xanthates | Ultra-fast |







