Natural rubber is an elastic material that is mainly obtained from the sap of hevea trees. East Asian countries, especially Malaysia and Indonesia, have extensive sources of this material due to their special climatic conditions. These trees also grow in some parts of South America, such as Brazil, which has a tropical climate. Some properties and characteristics of NR have led to the use of this material in various applications. The natural rubber has a very high resistance to tensile and tears, it can also be a good resistance material against light or heat. This natural material has very high adhesion to various metals, which has made this material very usable in the automotive industry very wide.
A Brief look at the History of Natural Rubber:
At the begging of World War I, some countries such as Germany and Japan were able to make some kind of rubber by using Butadiene, which of course, was not good in quality. In 1839, an American scientist invented a new method for curing rubber based on sulfur, which solved the problem of adhesion. Over time, in 1888, researchers in the rubber industry developed an additive that could Strengthen the properties of natural rubber and at the same time reduce its price, today this material is known as black carbon. During World War I, Production of this polymer ceased following the Japanese invasion of the tire industry, Therefore, the United States began producing synthetic rubber production units, which are so widespread today that 88% of used tires are of artificial origin in the United States.
What is the Structure of Natural Rubber?
Natural rubber structure is mainly long chain length and is a type of polyisoprene whose chemical structure is shown in the figure below:
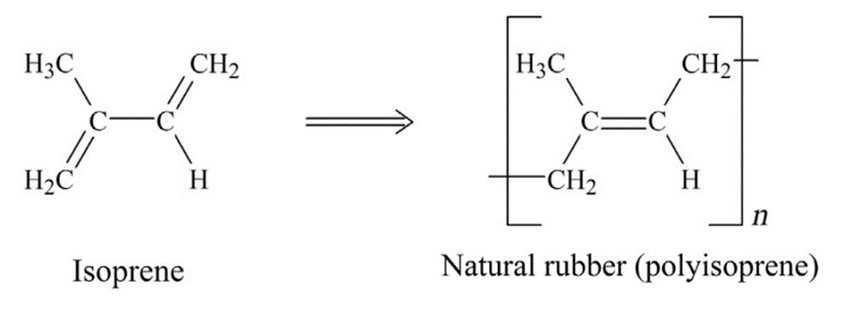
The monomers of this material are placed next to each other completely randomly, which makes the material reversible for this material.
How the Natural Rubber is Prepared?
As mentioned, the main source of natural rubber is Hevea trees. To produce natural rubber, grooves are first made on Hevea trees where the leachate comes out. The resulting leachates are then filtered by adding ammonia. Natural rubber is removed by an operation called coagulation. This process can be done by adding acids or some salts, natural rubber is separated from the liquid in the form of a white paste that needs to be dried. A newer method is to turn the coagulated leachate into granules by using rotating blades or cutting between two rollers that rotate at different speeds. The grains are then dried in mechanical dryers for several hours, in the old method they used air or wood smoke for drying, which took several days. In any case, the sheet or dried grain is dense and models are made weighing 33 kg.

The raw material does not show good physical and mechanical properties. The addition of sulfur increases the strength and tensile strength of the rubber. This process is called vulcanization. After vulcanization, fillers such as carbon dioxide are added to the rubber to increase its elasticity and strength. After these steps, the raw rubber becomes a tensile material with high physical and mechanical properties.
What are the Properties of Natural Rubber?
Compared to synthetic rubber, this material is cheaper and it also has good flexibility at low temperatures. This material has high water resistance, it is also a resistant polymer to impact. These properties have made NR widely found in various industries including the automotive industry.






