Description
Aluminum sulfate is a water-soluble compound known in the industry under the brand name 17% powder alum or 17% aluminum sulfate. One of the most important uses of white alum is drinking water and wastewater treatment, which can treat up to 99% of water impurities. Shanghai Chemex is one of the most reputable manufacturers of this chemical in the world. In this text, we try to acquaint you more with this non-flammable substance and its properties and applications.

Physical and Chemical Properties:
Aluminum sulfate is a white chemical with the molecular formula Al2 (SO4) 3, which is the second most widely used aluminum compound after aluminum oxide and appears as a liquid or crystalline powder and is often used with a purity of 17%; The chemical and physical properties of this mineral salt compound areas table follow:
| Chemical formula | Al2(SO4)3 |
| Molecular Weight(g/mol) | 342.15 |
| Appearance | Solid |
| odor | Odorless |
| Taste | Sweet taste |
| PH | 5.5-7.5 |
| Density (g/cm3) | 2.672 |
| Melting point (° C) | 770 |
| Color | White |
| Form | crystals, pieces, granules, or powder |
| Chemical Structure Depiction | 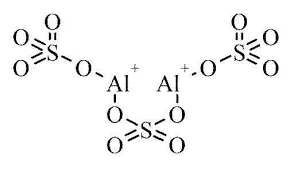 |
Synthesis and Production of Aluminum Sulfate:
Methods of producing aluminum sulfate can be divided into two general categories, which we will describe in the following:
- In the first method, this material can be obtained by reaction of sulfuric acid with waste materials of aluminum factories. The product of this process is a gray powder that contains a percentage of white alum with a relatively large number of impurities, in some industries such as zinc or carbon manufacturing industries that do not require high purity of this material, we can use this material without purification.
- In the second method, which is more common, water-diluted sulfuric acid is reacted with its hydroxide or its soil to produce this non-flammable material. This method usually has a high production speed and consumes less hydroxide, which is much more economical and it is also better than the first method in terms of quality.
Aluminum Sulfate Uses:
The applications of aluminum sulfate can be very wide, the most important of which are the following:
Agriculture:
The use of white alum in agricultural industries as agrochemical can significantly reduce soil acidity and make the soil of gardens and agricultural lands more fertile. This reduction in acidity is due to the hydrolysis of this salt and the formation of aluminum hydroxide precipitates and dilute sulfuric acid solution.
Some ornamental plant growers use this chemical to change the color of their flowers.
Water Treatment:
This mineral chemical is an important material in the water treatment industry that is used to clean, clarify and increase the water quality of pools and lakes. Similar products include polyacrylamide or sodium hypochlorite.

Aluminum sulfate is able to absorb impurities in water through electrical absorption and settle in the form of sludge. Then these contaminants can be easily removed from the bottom of the tank. Factors such as nontoxicity, no wide pH change, easy use, and cheap price have led to the expansion of aluminum sulfate consumption in the water treatment industry. This process is called coagulation and is used to remove water turbidity. This product, along with other chemicals such as hydrated lime and poly aluminum chloride, has a strong coagulation property for water purification.
Dyeing:
In the dyeing industry, it can play the role of a color stabilizer on the fabric.
Cosmetics:
Various forms of alum, such as potassium aluminum sulfate or ammonium aluminum sulfate, are used as active ingredients in the production of deodorants and antiperspirants. Of course, the US Food and Drug Administration has not accepted aluminum salts as an antiperspirant for many years, but it is still used in Asian countries.
Production of Fire Extinguishers:
If aluminum sulfate is heated to 580 to 900 ° C, it decomposes to alumina and sulfur trioxide, and when water is added to it, it forms hydrated salts.
Combining Al2 (SO4) 3 with baking soda (sodium bicarbonate) and foam stabilizer can produce carbon dioxide, which is used as a fire foam. Its reaction equation is as follows:
Al2(SO4)3 + 6NaHCO3 → 3Na2SO4 + 2Al(OH)3 + 6CO2
Other Uses:
Various compounds of aluminum sulfate can be used in the production of baking soda, waterproofing concrete, pencil production, painkillers to reduce insect bites, fungicides, leather production, and the production of aluminum compounds.
Buy Aluminum Sulfate:
For more information on purchasing and ordering this product through the numbers available on the site, please contact our experts in Shanghai Chemex.
Side Effects:
Aluminum sulfate is a substance with acidic properties that can cause itching, burning, or coughing if it comes in contact with the skin and eyes or inhales its vapors. Eating it can also cause severe burning in the stomach and gastrointestinal tract and cause nausea and vomiting.
First Aid :
To rescue a person whose concentrated fumes of aluminum sulfate have entered his lungs, the injured person should be transported to the open air. Also, if you swallow this substance, drink a glass of milk instead of water to dilute it. If aluminum sulfate comes in contact with the skin and causes chemical burns, you should take off your clothes and wash the affected area with plenty of cold water.
Packaging and Storage:
This non-toxic chemical should be stored in a cool, dry place in a multi-layered compartment so that it does not mix with other materials. The high temperature causes this chemical to lose its crystalline state and become leachate and the crystals to stick to each other.
Generally, the acidity of Aluminum sulfate is low and as a result, it has poor corrosion. Therefore, it is better than the pipes, fittings and all the containers that are in the vicinity of this material are made of plastic, fiberglass, stainless steel, or other resistant materials.







Reviews
There are no reviews yet.