Description
Etidronic acid, abbreviated as HEDP with the chemical formula C2H8O7P2, is one of the compounds of the Bisphosphonates family and is available in both solid and liquid forms. HEDP is formed by the reaction of phosphorus trichloride with acetic acid or by the reaction of a mixture of acetic acid / acetic anhydride with phosphoric acid.
This compound is known as a good and cost-effective corrosion inhibitor that is used in various industrial applications such as water treatment and detergents.

Physical and Chemical Properties:
Etidronic acid is an inorganic phosphoric acid corrosion inhibitor that forms stable compounds with iron, copper, and zinc ions.
Also, this organic compound shows good inhibitory effects of sediment and corrosion at temperatures below 250 ° C and has good stability at high pH. The compound is difficult to hydrolyze and decomposes difficult at high temperatures and direct light.
In the table below some of the chemical properties of this product are mentioned:
| Chemical formula | C2H8O7P2 |
| Molecular Weight(g/mol) | 206.03 |
| Appearance | Colorless or Light beige liquid |
| odor | odorless |
| Density (g/mL at 25 °C) | 1.4 |
| Melting point (° C) | 198-199 |
| Boiling point (° C) | 578.8 |
| Water Solubility (g/100 mL at 20 °C) | 1.5 |
| Other names | Etidronic acid, etidronate, HEDP,
1-Hydroxyethylidene-1,1-diphosphonic acid |
| Chemical Structure Depiction | 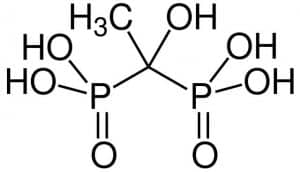 |
Synthesis of HEDP:
Etidronic acid (HEDP) can be synthesized in two ways:
The first method:
In this method, by hydrolysis of phosphorus trichloride and acetic acid, in the presence of the third type of amine as a catalyst, according to the following relationship, HEDP is obtained.
2PCl3+CH3COOH+5H2O ——–> HEDP + 6 HCl
The second method:
In this method, by hydrolysis of a mixture of acetic acid – acetic anhydride with phosphoric acid in the presence of nitric acid, hydroxyethane di phosphonic acid (HEDP) is obtained.
The first method of synthesizing etidronate has more advantages than the second method, which are mentioned below:
- The required raw materials are cheaper and available.
- Reactions take place at atmospheric pressure of 25 to 125 ° C. As a result, preparation is easier and test safety is increased.
- The purity of the final material is 97% and the yield is more than 95%.
- Due to economic considerations and the above, the first method is currently used more in industry.
Applications and Uses of Etidronate:
Etidronic acid has many uses, including; Cooling systems, industrial water treatment, industrial detergent production, stabilizing agent in hydrogen peroxide solution, separating agents in the textile industry, corrosion inhibitors, hardening agents, polishing materials, treatment, and medicine.
In the following, we will explain some of these many applications of HEDP:
Water treatment: One of the most important applications of this material in industries related to water and wastewater treatment. This material due to its anti-scaling properties by combining with metal ions such as calcium, iron, and copper can prevent groundwater contamination with these metals, In addition, this material prevents stains in swimming pools.

Medical: This substance is used to strengthen bones and treat osteoporosis. Bisphosphonates primarily reduce osteoclastic activity, which prevents osteoporosis and, in the long run, increases bone strength.
Unlike bisphosphonates, etidronate inhibits bone density. For this reason, other bisphosphonates, such as alendronate, are preferred in the fight against osteoporosis.
Cosmetic: Hydroxyethane diphosphonic acid can retain the preservatives in oxidized compounds by trapping heavy metal ions used in cosmetics, increasing the durability of cosmetics.
Although the use of hydroxyethane diphosphonic acid in cosmetics is not prohibited, warnings about the use of this substance in skin preservatives and soaps have been given.
Detergent industry: HEDP is an essential element in the detergent industry that have antimicrobial and bleaching properties. This chemical compound, in addition to stabilizing the hardness of water and the dispersion of contaminated particles, is able to remove stains.
Safety Information:
- May be corrosive to metals.
- Harmful if swallowed.
- Causes severe skin burns and eye damage.
- Toxic to aquatic life.



First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Store in a cool, dry, well-ventilated area away from heat, sparks and ignition sources, and incompatible materials.







Reviews
There are no reviews yet.