Description
4- Chloro-benzoic acid is an organic compound with the chemical formula ClC6H4CO2H. This organic compound is a mono chloro-benzoic acid that carries a chloro-substituent at position 4. It is the conjugate acid of 4- chloro-benzoates and is derived from benzoic acid. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

physical and Chemical Properties:
4 -Chloro-benzoic acid appears as triclinic crystals or light fluffy white powder and is soluble in some organic solvents. This compound is also incompatible with strong oxidizing agents and strong bases. The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | C7H5ClO2 |
| Molar mass(g/mol) | 156.57 |
| Appearance | white solid |
| Density(g/cm3 ) | 1.571 |
| odor | odorless |
| Log P | 2.65 |
| P ka(at 25 °C) | 3.98 |
| Melting Point(° C) | 241.5 |
| Boiling point(° C) | 276 |
| Solubility | Insoluble in, benzene, and carbon tetrachloride;
soluble in ethanol, water, chloroform |
| Color | white |
| Form | triclinic crystals |
| Synonyms | p-Chloro- benzoic acid
para-Chloro- benzoic acid |
| Chemical Structure Depiction | 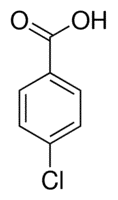 |
Formula and Structure of 4-Chlorobenzoic Acid:
The structure of this compound with the chemical formula ClC6H4CO2H includes the arrangement of atoms and the chemical bonds that hold the atoms together. This organic molecule includes a six-membered ring, a carboxylic acid, and a hydroxyl group. These compounds are benzoic acids that carry the halogen atom on the benzene ring.
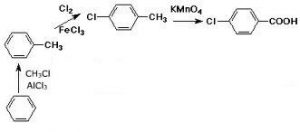
4-Chlorobenzoic Acid Production Process:
The preparation of p-chlorobenzoic acid from a mixture of ortho and parachlorotoluene, involves the oxidation of this mixture using oxygen-containing gas in a reaction medium consisting of monocarboxylic acid.
4-Chlorobenzoic Acid Uses:
- prevents the growth of fungi, yeasts, and some bacteria. It is used as a pH regulator and preservative in food and the growth of microbes is used to keep food safe.
- Used as agrochemical in agriculture to repel pests and insects.

- As a laboratory reagent in the manufacture of chemicals.
- preservative for adhesives and paints.
- 4- chloro-benzoic acids are also used in the manufacture of medicines.
Safety Information:
Irritating to eyes, skin, mucous membranes, and upper respiratory tract.



First-aid measures:
Skin Contact: Immediately flush skin with water.
Inhalation: move the person to the fresh air.
Eye Contact: Rinse eyes with water for several minutes.
Ingestion: Rinse mouth. Do not induce vomiting.
Packing and storage:
Keep the container tightly closed in a dry and well-ventilated place.






Reviews
There are no reviews yet.