Description
Hydroquinone is an aromatic organic compound in which two hydroxyl groups are attached to benzene rings in ortho and para positions. This compound was named in 1843 by a man named Friedrich Wehler. Widely used in cosmetics as a skin brightener and clarifier. This compound reduces the rate of melanin formation by inhibiting the enzyme tyrosinase. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world

Physical and Chemical Properties:
This antioxidant appears as white and odorless granules. Phenol is an aromatic chemical compound available naturally and synthetically. The most important physical and chemical properties of this hydroxy compound can be summarized in the following table:
|
Chemical formula |
C6H6O2 |
| Molecular Weight(g/mol) | 110.112 |
| Appearance | white solid |
| odor | Odorless |
| taste | A slightly bitter taste in aqueous solutions |
| Density (g/cm3) | 1.3 |
| Melting point (° C) | 172 (342 °F; 445 K) |
| Boiling point (° C) | 287 (549 °F; 560 K) |
| Water Solubility (g/100 mL at 15° C) | 5.9 |
| color | White |
| form | crystals |
| Flash Point (° C) | 165 (329 °F) |
| Vapor Pressure (mm Hg at 302 °F) | 4 |
| Chemical Structure Depiction | 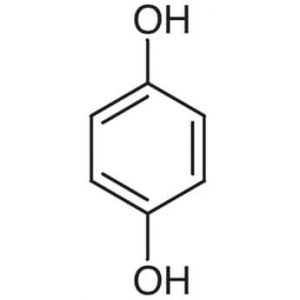 |
Production Process of Hydroquinone:
This phenol is present in some plants and fungi, but using natural resources to produce this chemical is a relatively difficult operation, and a large volume of this hydroxy compound is produced in the laboratory. To produce this aromatic compound, we can also be produced through three chemical processes, including oxidation, reduction, and alkylation reactions. Benzene or phenol are usually used to produce hydroquinone, and during a redox process, we can synthesize the desired substance.
There are three current production processes for Hydroquinone:
- Phenol hydroxylation;
- Oxidation of aniline to quinone in the presence of manganese dioxide and sulfuric acid;
- Synthesis of hydroquinone from acetylene and pentacarbonyl iron.
Hydroquinone Uses:
This compound is used in various industries, the following are the most important applications:
- In the photography and black and white photography industry, a combination of hydroquinone with Phenindione is used.
- Hydroquinone, as a polymerization inhibitor, uses its antioxidant properties to prevent the polymerization of acrylic acid, methyl methacrylate, cyanoacrylate, and other monomers that are prone to radical polymerization.
- Hydroquinone; as a free radical scavenger, increases the shelf life of light-sensitive resins.
- It is used as a chemical intermediate in the synthesis of pharmaceuticals, chemicals, and agricultural chemicals.
- In the paint, ink, and, adhesive industries.
- Used as an antioxidant in the food industry.
- Corrosion in steam-generating systems causes many problems. Hydroquinone helps to solve this problem by reducing dissolved oxygen in the inlet flows to the boilers.
- This phenol is also used in the rubber and plastic manufacturing industries.
- This aromatic compound is used in the formulation of most cosmetics
- In the production processes of polystyrene as a photochemical substance.
- In the production of engine oils.
- Inhibitory role in the production of vinyl acetate and acrylic monomers.
Can Hydroquinone be used in Cosmetics?
The use of this aromatic compound is very widespread in the cosmetics industry and is mainly used in the production of gels, ointment, moisturizing creams, and leucine. This hydroxy composition has anti-spot and clearing properties for the skin and helps to lighten the skin. However, it may cause discomfort to some sensitive and dry skin. Hydroquinone ointment may be present in concentrations of 15% and 20% or more in products made with it, but according to international standards, the use of products with concentrations higher than 2% requires a prescription. In some cosmetics hygienic 4% hydroquinone is used to remove brown spots.
Safety Information:
- Hydroquinone creams can contain a substance called sodium sulfite, which can cause allergies in some cases. It is better to do allergies before using this product before use to make sure;
- Exposure to sunlight after using hydroquinone can cause toxicity and side effects;
- Symptoms of hydroquinone poisoning;
- Feeling of suffocation, vomiting, muscle strain, headache, seizures, shortness of breath, coma, and respiratory failure.
Packing and Storage:
Keep the container tightly closed in a dry and well-ventilated place.








Reviews
There are no reviews yet.