Description
Linear alkylbenzene sulfonic acid, which is briefly known as LABSA, is a member of the anionic surfactant family. Surfactants are able to break down the binding between the surface and lipids. So it removes strong stains and fats because of its lipophilic property. Generally, this substance is the cheapest common surfactant used in the powder and liquid detergent industries. It is also used as a catalyst.
How to buy LABSA?
Shanghai Chemex is one of the suppliers of sulfonic acid in China. If you would like to know any further information about linear alkylbenzene sulfonic acid’s price or buying this item, our team will be delighted to help you with that.
Physical and Chemical Properties
Linear alkylbenzene sulfonic acids chemical formula is CH3(CH2)11C6H4SO3H. It appears as a thick, brown liquid. The most important physical and chemical properties of this material are summarized in the following table:
| Chemical Structure Depiction | 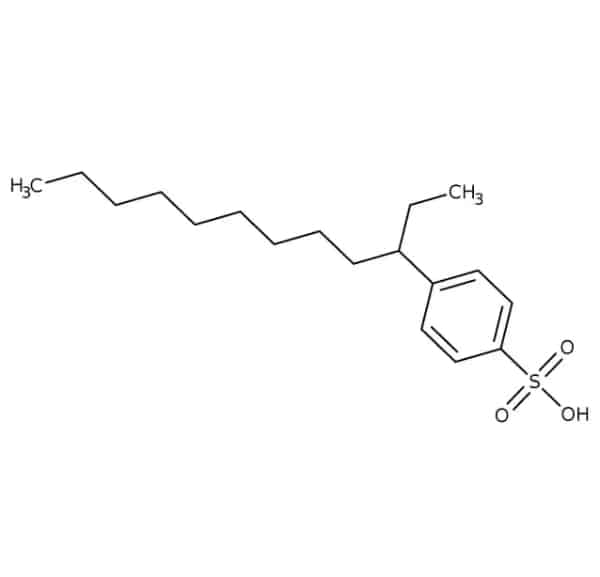 |
| Name | Linear Alkyl Benzene Sulphonic Acid |
| Molecular weight | 326.49 g/mol |
| Appearance | Brown liquid |
| Density | 863 kg/cm3 |
| Boiling point | 315 °C |
| Melting point | 10 °C |
| Solubility | Soluble in water and organic solvents |
LABSA consists of a hydrophilic sulfonate and a hydrophobic alkylbenzene group.
Linear alkyl benzene sulfonic acid uses
The most important application of this acid is in the production of detergents. Industries use LABSA in dishwashing liquid, laundry powder, cleaning powders, liquid and oil soaps, and also household detergents.
Linear alkylbenzene sulfonate mode of action
This chemical substance is a common surfactant. The word “surfactant” comes from surface-active agents. Surfactant molecules have two ends. One of them likes water molecules (hydrophilic), and the other hates them (hydrophobic), and that is why, when they are in the water, they create beads. The outside of the beads has water-loving ends, and on the inside, it has water-hating ends to keep itself away from water molecules.
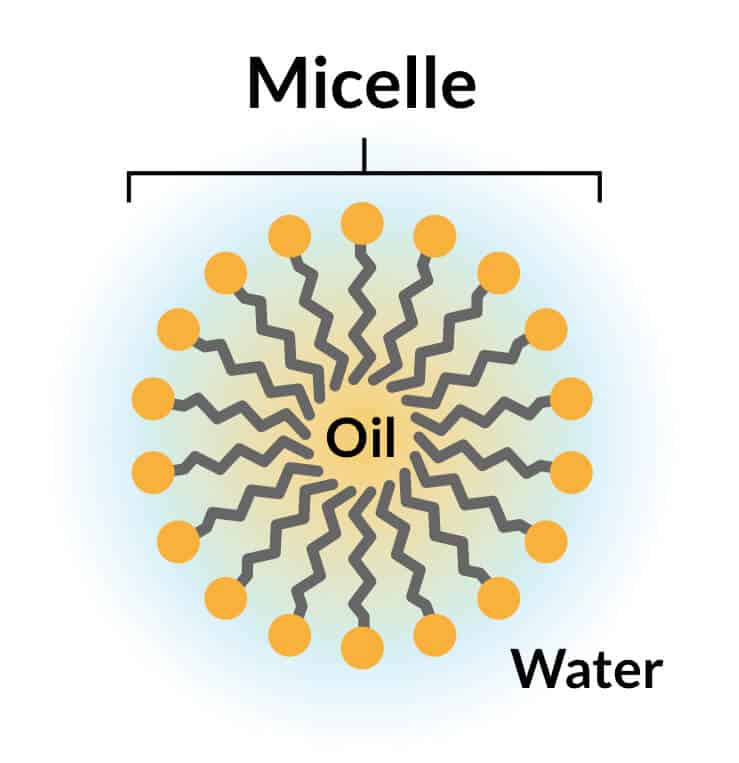
When we add Linear alkylbenzene sulfonic acid anionic surfactant to the detergent like soap and use water, the surfactant hydrophobic ends of surfactant molecules want to attach anything to the water. as a result, it surrounds dirt and fats on the area and floats them in the soap solution.
- Paint industries also use LABSA, most of the washable dyes are either sulfonic acid or have sulfonyl agents in their structure.
- It is used in the pharmaceutical industry as an important and practical factor. For example, they play an important role in the production of antibacterial drugs and infection treatment medicines.
Linear alkylbenzene sulfonate side effects
This substance is one of the relatively dangerous organic acids. For this reason, we have given you the following tips so that you are more aware of this and act with more complete information and more caution:
- Eye contact can cause severe eye irritation. In the event of such an accident, keep your eyes open and rinse with lukewarm water for 15 minutes.
- Also, if the skin of your hand comes in contact with sulfonic acid, wash the contact area immediately with plenty of soap and water and go to a medical center as soon as possible.
- Inhalation of its vapors stimulates the respiratory system, so the injured person is transported to the open air, and if the breathing problem persists, the oxygen device should be connected.
If any of this material is spilled on the ground and spread in the warehouse environment, collect it immediately and transport it to the chemical waste. You should store packages containing LABSA in a dry, cool, well-ventilated area and keep them away from sources of heat.
The bottom line
Linear alkylbenzene sulfonate is an anionic surfactant. It can surround impurities and flow them in the solution. Also, Industries use this material in some paints and producing antibacterial drugs. However, you should pay attention to store it in a cool place and keep it away from heat.
Frequently asked question
What is the use of linear alkylbenzene sulfonate?
Alkylbenzene sulfonate is another name for Linear alkylbenzene sulfonate. So as LABS, this compounds can be used in detergents formulation and washable dyes.
Why LABSA is used in detergent?
Because it is a surfactant and the role of surfactant is to surround dirt on the area and floats them in the soap solution. It is high soluble in water too.
What is the use of LABSA?
They are widely used in washable paints, soaps, laundry and dishwashers formulation.
Can alkyl benzene sulfonate be used as detergent?
Yes, alkyl benzene sulfonate is an anionic surfactant which can remove dirt and fats from the surface.






lina –
Is LABSA toxic?
china chemicals –
Studies have shown that linear alkylbenzene sulfonate (LABSA), a type of omnipresent pollutant, can cause toxic damage to aquatic plants.
Ella –
Is this surfactant ionic?
china chemicals –
Yes, LABSA is an ionic surfactant.