Description
Oxalic acid, also known as ethanedioic acid, is a strong, organic acid that is found naturally in many plants, such as spinach, ginger, and chocolate and is synthesized synthetically in laboratories. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
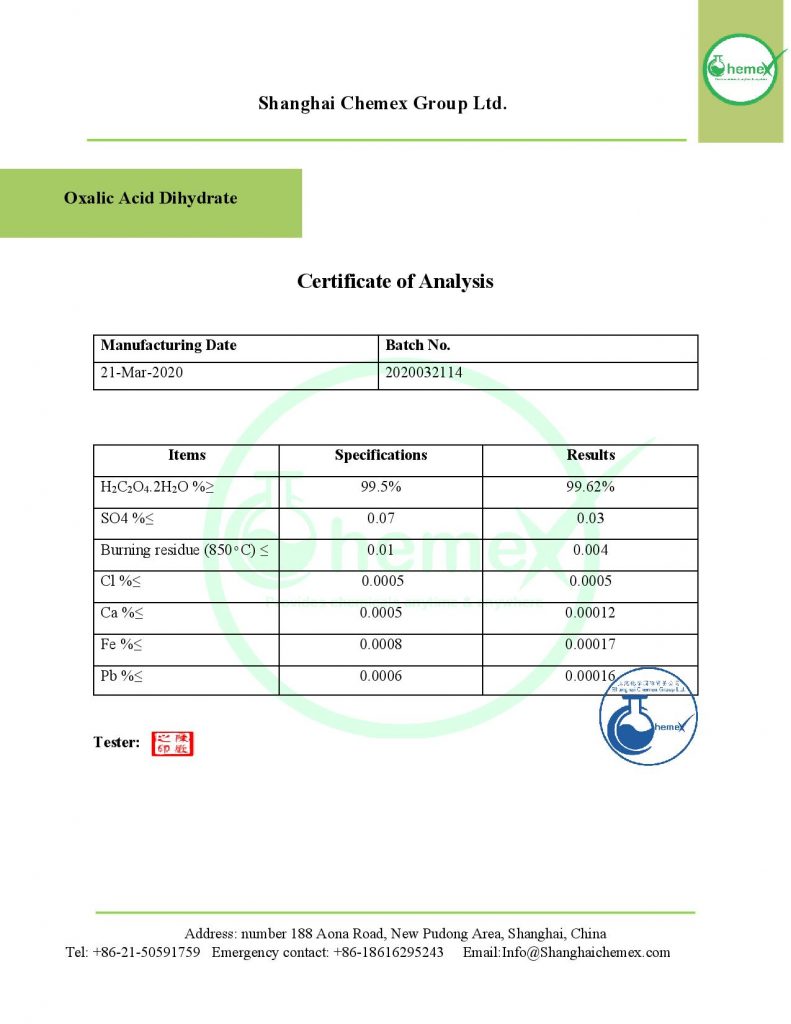
This is an organic acid is available in the market in both anhydrous and aqueous forms. Its aqueous form has two molecules of water per molecule of this substance in its vicinity, but its anhydrous form is crystalline and white. The aqueous form of this substance loses its water if exposed to 100 ° C; In the table below some of the chemical and physical properties of this product are mentioned:
| Chemical formula | C2H2O4 |
| Molecular Weight(g/mol) | 90.034 |
| Appearance | solid |
| odor | odorless |
| Density (g/cm3) | 1.90 |
| Melting point (° C) | 189 to 191 |
| Color | White |
| Form | powder or granular |
| Synonyms |
oxalic acid, ethanedioic acid, Aktisal, Aquisal |
| Chemical Structure Depiction | 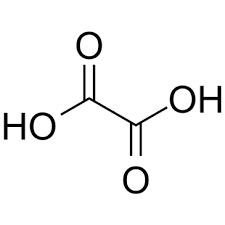 |
The Production Process of Oxalic Acid:
- In general, oxalic acid can be synthesized from the oxidation reaction of carbohydrates or glucose using nitric acid or air in the presence of a vanadium pentoxide catalyst.
- Another way to produce this substance is to prepare the diester by the carbonylation reaction of alcohols and the hydrolysis of the diester to oxalic acid.
4 ROH + 4 CO + O2 → 2 (CO2R)2 + 2 H2O
- It can be prepared in the laboratory by oxidation of sucrose using nitric acid in the presence of a small amount of vanadium pentoxide as a catalyst.
Oxalic Acid Uses:
- In wastewater treatment to remove calcium deposits
- As a stabilizing factor in the dyeing process
- Due to its high acidity as a reducing agent
- As a diluent and purifying agent in the pharmaceutical industry.
- To sterilize medical equipment and as a reducing agent in drugs and antibiotics
- To eliminate a specific pest in agriculture. like Phosphorous Acid
- In the synthesis of organic materials such as cobalt oxalate, nickel oxalate, and pentaerythritol
- As an anticoagulant for laboratory blood samples.
- Electroplating agent and surface modification and coating
- In the formulation of detergent products
- In the plastics industry for the production of polyvinyl chloride, amino plastics, urea-formaldehyde plastics
Is Oxalic Acid Good for Cleaning?
Oxalic acid is an ideal product for cleaning and bleaching, and among its applications:
- This acid, along with citric acid and tartaric acid, is used to remove some stains.
- The bleaching properties of this material are used to sterilize household appliances.
- It can also be used to remove iron rust.
- 5% solution of this acid is used to remove ink stains.
- It is very useful for removing dirt from surfaces.

Buy Oxalic Acid:
Contact our experts in Shanghai Chemex to buy oxalic acid at the best price and quality.
The global market for Oxalic Acid:
China can be considered the largest exporter and producer of this chemical in the world, it exports more than 25% of its oxalic acid to countries such as the United States, Asian countries such as Japan and Indonesia, as well as parts of Europe. The use of this acid in textiles, metal preparation, manufacturing of chemicals and pharmaceuticals, and leather industries, and… increased the sales of oxalic acid in global markets. It is interesting to know that in 2016, the largest volume of oxalic acid purchases was related to the pharmaceutical industry and is expected to maintain its position by 2021 with an annual growth of 3.4%.
Safety Information:
This acid is flammable in its pure state and catches fire if it is in a hot or near heat environment. It is in the category of corrosive compounds and in contact with the skin, eyes or if swallowed, it can irritate the skin, and eyes and cause poisoning.
First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.


Handling and Storage:
Keep the container tightly closed in a dry and well-ventilated place.






Maryam –
What foods contain oxalic acid?
china chemicals –
Oxalic acid occurs naturally in many plants like the following:
Fruits
Cocoa
Leafy green vegetables
Nuts
Spinach
Sweet potatoes
Star fruit