Description
Potassium citrate is a derivative of citric acid that has the chemical formula K3C6H5O7. This compound is added to foods to regulate acidity. Potassium citrate is produced by the addition of potassium carbonate to citric acid. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
Potassium citrate is a white powder with, an odorless, salty taste and absorbent. It is soluble in water and glycerin and insoluble in ethanol. The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | K3C6H5O7 |
| Molar mass(g/mol) | 306.395 |
| Appearance | solid |
| Density(g/cm3 ) | 1.98 |
| odor | odorless |
| Taste | Cooling, salty taste |
| Melting Point(° C) | 180 |
| Boiling point(° C) | 230 |
| Solubility in water | soluble |
| Solubility | soluble in glycerin
insoluble in ethanol |
| PH | Between 7.5 and 9.0, about 8.5 |
| Color | white |
| Form | Crystals, granules, or powders |
| Synonyms
|
Tripotassium 2-hydroxy propane-1,2,3-tricarboxylate |
| Chemical Structure Depiction | 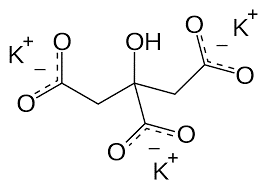 |
Production Process of Potassium Citrate:
Citric acid is first produced using a fermentation process. After fermentation, more processing is required to separate the by-products and purify the citric acid. The addition of potassium hydroxide to citric acid leads to the formation of potassium citrate crystals. These crystals are separated from the solution and used as a food additive.
Potassium Citrate Uses:
- Oral potassium citrate is rapidly absorbed by the body and excreted in the urine. Because it is an alkaline salt, it is effective in reducing urination or acidification of urine and reducing pain.
- It is also used as an alkaline substance in the treatment of mild urinary tract infections such as cystitis.
- This compound is used as a food additive, potassium citrate can improve heart rate and bone health.
- Potassium citrate is an effective way to treat and control gout and arrhythmias.
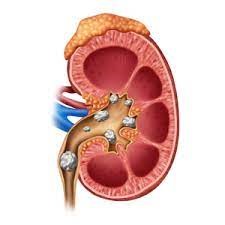
- It is widely used to treat kidney stones and is often used by patients with cystinuria.
- It is also used in child care products, hair dyes, soaps and detergents, cosmetics, and skincare products.
- This chemical is also used as a cleaning agent in industries.
- This substance is used with sodium to regulate the amount of water in the body and help blood pressure in the body.
- One of the most important causes of stroke is high blood pressure. It lowers blood pressure and thus reduces the risk of stroke.
Is Potassium Citrate in Food Safe?
Natural potassium citrate is found in high-fiber foods such as vegetables, fruits, and nuts. If you use enough of these natural substances then your body will receive enough potassium. It has an alkaline effect on the body and helps regulate PH. This compound is used as a food additive and can improve heart rate and bone health, as well as help with kidney stones.

Can Potassium Citrate dissolve Kidney Stones?
Potassium citrate is often used to treat kidney stones. In particular, it helps remove protons or positively charged atoms, which reduce acidity in the urine. In addition, it can crystallize rock-forming salts such as calcium oxalate, calcium phosphate, and uric acid in the bladder. This raises the pH of the urine and makes it alkaline. In addition, it increases the amount of citrate in the urine and thus reduces the risk of stone formation. In general, maintaining the acid balance in the bladder prevents the formation of kidney stones.
Buy Potassium Citrate:
Potassium Citrate is ready to be delivered to you, dear customers, in 25 kg bags. Shanghai Chemex is ready to receive the orders of our dear customers, provide the required Potassium Citrate product with the best price and quality, and send it as soon as possible by a reliable transportation system.
Safety Information:
Pharmaceutical forms of potassium citrate are usually prescribed in the form of tablets and dilute aqueous solution (potassium citrate syrup). Side effects of potassium citrate medications can include irregular heartbeat, muscle weakness, severe stomach pain, abdominal discomfort, nausea, vomiting, diarrhea, elevated potassium levels, and rarely cardiac arrest. If you think you have used too much potassium citrate, please pay attention to seek medical information.
Packing and storage:
Store the medicine in a closed container at room temperature, away from heat, moisture, and direct light.






Reviews
There are no reviews yet.