Description
Sodium saccharin, or benzoyl sulfimide, is a synthetic, non-nutritive sweetener as well as a white crystalline solid. It is about 300 to 400 times sweeter than sucrose, but has a bitter or metallic taste, especially at high concentrations. Saccharin has no energy or nutritional value; Therefore, it does not pose a risk to people with diabetes. This compound is commonly used as a sugar substitute because it does not contain calories or carbohydrates. This substance is not broken down in the human body, so it is excreted without changing the body. Shanghai Chemex offers this high-quality chemical very safely.

Physical and Chemical Properties of Sodium Saccharin:
Sodium saccharin is in the form of odorless white crystals or white powder. Its aqueous solution in the presence of litmus paper shows a neutral or alkaline property, but the presence of phenolphthalein does not show this alkaline property. When exposed to air, it loses its moisture and has a very sweet taste. This compound is about 300 times sweeter than sugar and is widely used as an artificial sweetener and, like many other salts, decomposes into its constituents when dissolved in water; In general, the most important physical and chemical properties of sodium saccharin can be summarized in the following table:
| Name | Sodium saccharin |
| Chemical formula | C7H4NNaO3S |
| Molecular weight (g/mol) | 206.17 |
| Melting point (°C) | >300 |
| Boiling point (°C) | 438.9 |
| Solubility | Soluble in water, slightly soluble in ethanol |
| Chemical Structure Depiction | 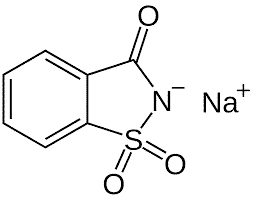 |
The Production Process:
Sulfonation is used to produce sodium saccharin. In the first stage, toluene is sulfonated with chlorosulfonic acid and the final product undergoes an amino process in the presence of ammonia. In the last step and during the oxidation process of ortho-toluene sulfonic acid, the final product can be obtained.

Sodium Saccharin Uses:
In general, many industries use this chemical, Food industry, chemical and agricultural industry, plating industry, and pharmaceutical are the most important industries that use this synthetic material. In the following, we will become more familiar with the applications of this material.
Food:
This food additive is used in the production of food supplements, sauces, jams, bread and coffee industries, and beverages. The sweetening of baked goods, cookies and muffins, foods, and chemical drinks are common uses of this substance.
Plating:
In the plating industry, sodium saccharin is used as an additive in metalworking liquids and adhesives. For example nickel plating. Saccharin is used as an electrolyte. Plating is a method in which an electric current is applied through a solution until the surface of another metal is covered with another metal. The nickel plating industry using sodium saccharin is very popular and sodium saccharin plating is also used in gold and palladium plating baths. like Propargyl Alcohol Propoxylate

Agriculture:
Utilize in agricultural industries to produce pesticides and herbicides.
Cosmetic Products:
Sodium saccharin is used in the manufacture of personal care products or health products such as toothpaste, and mouthwash, in addition, the use of this chemical in the production of cosmetics is also very common.
Pharmaceutical:
In pharmacy, this salt is used in the formulation of multivitamin tablets and chewable tablets. This improves the taste quality of pharmaceutical products.
Sodium Saccharin in the Production of Toothpaste:
Sodium saccharin is used to improve the taste of toothpaste and sweeten its taste. Because this compound is so sweet, very small amounts of it can improve the taste of toothpaste.
According to studies, this toothpaste additive has no health benefits and is merely a sweetener used to improve the taste of toothpaste.

Buy Sodium Saccharin:
To buy sodium saccharin, contact Shanghai Chemex experts and get this product with the best price and guaranteed quality in the fastest possible time.
Side Effect:
Some people may be allergic to this substance, and taking saccharin-containing substances may be dangerous for infants, or in rare cases may cause musculoskeletal disorders. Excessive consumption may cause weight gain in some people, in general, monitor the consumption of this chemical in your diet, especially in children, and avoid overdosing.
Transporting and Storage:
Store this chemical in a cool, dry, well-ventilated place away from direct sunlight or heat.






LIDA –
Is sodium saccharin the same as aspartame?
china chemicals –
Aspartame, one of the most common artificial sweeteners, is a combination of two amino acids — phenylalanine and aspartic acid. Aspartame is 200 times sweeter than sugar and, like saccharin, contains no calories.