Description
Ammonium persulfate, or Ammonium deoxy sulfate, known by its chemical formula (NH4) 2S2O8, is an inorganic compound that is widely used in the chemistry of polymers, detergents, and bleaches. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.
Physical and Chemical Properties:
Ammonium persulfate, which is found as a colorless salt or white powder, like all peroxide sulfates (ammonium, potassium, and sodium), is produced by electrolytic processes and, of course, has a high solubility in water. This compound does not burn easily but may burn other organic matter. It is worth noting that ammonium persulfate releases toxic gases including nitric oxide, sulfur oxide, and ammonia when heated; The physical and chemical properties of this material can be summarized in the table below:
| Chemical formula | (NH4)2S2O8 |
| Molecular Weight(g/mol) | 228.18 |
| Appearance | solid |
| odor | Odorless |
| Density (g/cm3 at 25 °C) | 1.98 |
| Melting point (° C) | 120 (248 °F; 393 K) |
| Solubility | Freely soluble in water |
| Color | Colorless or white |
| Form | crystals or granular powder |
| Chemical Structure Depiction | 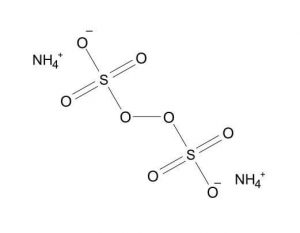 |
Ammonium Persulfate Preparation:
Ammonium persulfate was first produced by Hugh Marshall in this way:
Ammonium persulfate is prepared by electrolysis of a concentrated solution of ammonium sulfate or ammonium bisulfate in concentrated sulfuric acid with a high flow density. This method uses platinum electrodes. In the electrolysis process, this compound first loses HSO4, then at the anode, it produces acidic peroxy sulfate, The acid then reacts with ammonium sulfate to separate ammonium sulfate and ammonium persulfate by filtration when the contents reach a certain concentration in the anode.
(NH4) 2S2O8 + H2S2O8 → (NH4) 2S2O8 + H2SO4
Ammonium Persulfate Uses:
Due to its oxidizing properties, this compound is used in various industries. In the following, we will mention some of its applications:
- In the production of detergents such as soaps
- As a preservative in the wood industry
- In the production of cosmetic products such as exfoliators
- As a food preservative and acidity regulator in the food industry
- In the preparation of important commercial polymers such as styrene rubber, butadiene, polytetrafluoroethylene
- Production of paint and paper
- In the agricultural industry as fertilizer. like manganese sulfate monohydrate
- In the oil industry as a corrosive material in cutting incandescent metals and oil extraction
- Copper engraving on printed circuit boards
What is the use of Ammonium Persulfate in Agriculture?
One of the most important and main applications of ammonium persulfate as fertilizer in the agricultural industry. The reason for using this material as fertilizer is to improve the soil and help the plant grow better. The substance we put in the soil releases ammonium. Ammonium makes the soil more acidic, or in other words, lowers its PH. This decrease in PH provides the nitrogen needed by the plant and as a result, it grows better. Most of the soils used for plants are alkaline and calcareous, ammonium persulfate has a great effect on its improvement.

Buy Ammonium Persulfate:
For more information on purchasing and ordering this product through the numbers available on the site, please contact our experts in Shanghai Chemex.
Safety Information:
This salt, like other chemical compounds, has its own dangers. Direct contact of ammonium persulfate with the skin causes itching, inflammation, and pain. Also, inhalation can cause a sore throat and respiratory problems, so working with this substance requires all safety and health tips, including the use of gloves, goggles, and Special clothing. This material is explosive, high temperatures, and exposure to high heat and fire and also its combination with phosphorus and sulfur can cause it to explode.

First-aid measures:
- Eye Contact: In case of contact with the eyes, immediately flush the eyes with plenty of water.
- Skin Contact: Remove this material from the skin with plenty of soap and water.
- Inhalation: Remove to fresh air immediately. If not breathing, give artificial respiration.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
The storage environment of this compound should be dry and clean and well ventilated and away from fire, heat, and direct sunlight. The packaging must be sealed and have clear and sound labels.







Reviews
There are no reviews yet.