Description
Chlorobenzaldehyde, as a derivative of benzaldehyde, is obtained by the hydrolysis reaction of chlorotoluene. This compound is mostly used in medical and pharmaceutical industries as well as paint industries. But the main application of OCB is in electrical plating like Sodium allyl sulfonate (SAS). Chlorobenzaldehyde is very useful in killing bacteria and the use of a solution of this substance in cleaning and removing the body of various animals from infections, bacteria and is very common. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world; In the following, we will learn more about the main uses of Chlorobenzaldehyde and its properties and characteristics.

Physical and Chemical Properties of OCB:
The appearance of Chlorobenzaldehyde product can be a colorless liquid or a brown or yellow liquid. It is chemically water absorbent and direct light can affect its properties and characteristics. This colorless liquid is most used in biotechnology and electrical plating applications. The most prominent properties and characteristics of Chlorobenzaldehyde can be summarized in the following table:
| Name | OCB |
| Chemical formula | C7H5ClO |
| Molecular weight (g/mol) | 140.56 |
| Appearance |
Colorless to yellowish liquid |
| Density (g/cm3) | 1.248 |
| Boiling point (°C) | 212 |
| PH | 2.9 |
| Chemical Structure Depiction | 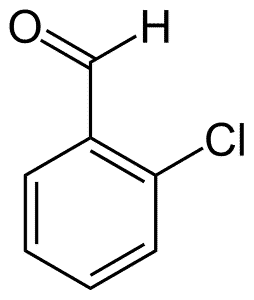 |
Synthesis and Production Process of Chlorobenzaldehyde:
In general, the production of Chlorobenzaldehyde can be done by the hydrolysis process. Chlorobenzyl chloride and chlorotoluene are substances that can be hydrolyzed to achieve the desired product. The OCB synthesis process can be performed with ortho or para chlorotoluene materials. This product is produced by chlorination of chloride chains or side branches with a relatively specific ratio and by performing a hydrolysis process on them.
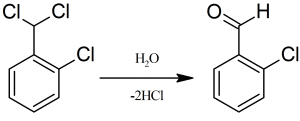
Chlorobenzaldehyde Uses:
The Chlorobenzaldehyde compound is one of the most practical and important substances used in the production of a wide range of alcohols.
- Due to its very good properties to prevent corrosion of metals, it is widely used in descaling, cleaning, and polishing metal surfaces.
- The pharmaceutical industry is another industry that specifically uses Chlorobenzaldehyde. More precisely, this liquid is used in the production of antibiotics.

In the following, we will briefly discuss the most important industries and applications of this chemical product:
- In agricultural industries, it is used to produce chemical fertilizers used on agricultural lands or to produce pesticides and herbicides.
- Dyeing and textile industries
- Metal plating and more precisely electric plating, nickel plating process, and some other metals are done with the help of this product.
Buy Chlorobenzaldehyde:
For more information on purchasing and ordering this product, please contact our experts in Shanghai Chemex through the numbers on the site.
Safety Information:
OCB is harmful to human health in the long run, it is important to observe safety issues when working with it, and be sure to use gloves, goggles, and special clothing. Avoid direct contact of this substance with body parts, especially eyes and skin, if some of this substance enters a person’s eye, be sure to rinse with water for 10 to 15 minutes, in case of contact with concentrated vapors, be sure to move the person to the fresh air and in more acute cases, go to medical centers or emergency.
Packing and Storage:
The sensitivity of OCB to light and high heat and its chemical tendency to react with oxygen has made the issue of storage very important. Use cold and dry places to store this material and try to pack them in such a way that they do not come into contact with oxygen in the air.







Reviews
There are no reviews yet.