Description
From the oxidation of benzene or a hydrocarbon in the presence of a vanadium oxide catalyst, maleic anhydride is produced.
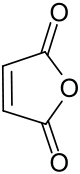
It is a white or colorless solid with a pungent odor that is produced on a large scale for industry for coatings and polymers.
Properties:
| formula | C4H2O3 |
| Molar mass | 98.057 g/mol |
| Appearance | White crystals or needles |
| Density | 1.48 g/cm3 |
| Melting point | 52.8 °C |
| Boiling point | 202 °C |
| solubility | In water |
Production of Maleic anhydride:
This compound can be made through butane vapor phase oxidation. In fact, traditionally, this compound can be produced from the oxidation of benzene or other aromatic compounds. In the continuation of the process, benzene or butane enters the hot air stream and the mixture passes through the catalyst bed at high temperature. The air ratio is controlled to prevent ignition. The catalysts used for butane and benzene are different; For benzene, they use vanadium pentoxide and molybdenum trioxide, and for butane, they use vanadium phosphate as a catalyst. In the traditional method, the use of benzene was no longer economical due to the increase in the price of benzene.
Applications of maleic anhydride:
This substance is present in a wide range of industries, which we will examine further.
Plastic and resin:
Almost 50% of the production of this material in the world use to make unsaturated polyester resins. These compounds are used in a wide variety of industries such as the construction of yachts, bathroom fixtures, automobiles, tanks and pipes. Also, this compound is hydrogenated to BDO, which is used in the production of thermoplastic polyurethanes and resins.
Treatment factors:
This compound is used as a curing agent in epoxy resins. Also, this material is used as an additive for lubricating oil related to diesel and gasoline engines as a corrosion inhibitor and dispersant.
Other uses:
This material also has many small applications, which include the following.
- In the food industry, malic acid, which is a derivative of maleic anhydride, use in artificial sweeteners and flavor enhancers.
- It use in personal care products such as hair sprays, glues and glosses.
- It is a precursor of compounds use for water purification detergents, insecticides, fungicides, medicines, etc.
This organic compound is always use in various industries.
MSDS:
Maleic anhydride is a toxic, dangerous and corrosive substance; Read the safety tips before working.
- Use gloves, goggles and protective clothing while working.
- Contact of this compound with the skin causes an allergic reaction and burns the skin; After contact, wash the area with plenty of water.
- Its contact with the eyes causes serious damage to them; After contact, wash the affected eye with water for a few minutes.
- Swallowing this substance causes acute toxicity, after swallowing drink one to two glasses of water and do not try to expel it by vomiting.
- After inhaling this dangerous substance, leave the work environment and breathe fresh air.
- Use water spray, carbon dioxide, resistant alcohol foam and dry chemicals to dry the fire caused by this material.
- Store this mixture in a closed container in a dry and ventilated place.
Avoid the dangers caused by this substance by following the safety tips.
Conclusion:
Maleic anhydride is an organic compound that is use in a wide range of industries such as resin manufacturing, food and chemical industries. This substance is toxic, dangerous and corrosive, and it is necessary to read and observe the safety tips before working with it.






Reviews
There are no reviews yet.