Description
Tetrahydrofuran (THF) is also known as butylene oxide and diethylene oxide, which is one of the most widely used chemical solvents used in various industries. This chemical compound is actually a heterocyclic organic compound consisting of 5 carbon atoms and one oxygen atom in a ring, usually in the form of a low viscosity liquid.THF is in the category of Industrial chemicals of Shanghai Chemex group Ltd.

This material is used to make elastic polyurethane fibers and high-consumption solvents in the PVC industry. It is also used in resin manufacturing industries. A similar solvent to this substance is Benzene.
Physical and Chemical Properties:
THF is a clear, colorless, volatile liquid that can be mixed with water. The chemical formula of this compound is C4H8O and it has a low viscosity. This substance is produced both in liquid and dry form; The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | C4H8O |
| Molecular Weight(g/mol) | 72.107 |
| Appearance | Colorless liquid |
| odor | Ether-like odor |
| Taste | Pungent taste |
| Density (g/cm3 at 20 °C) | 0.8876 |
| Melting point (° C) | −108.4 |
| Boiling Point (° C) | 66 |
| Solubility | Very soluble in acetone, benzene, ether, ethanol, and chloroform |
| Color | Colorless |
| Form | liquid |
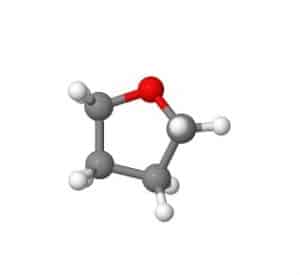
THF structure:
The chemical structure of this compound consists of 4 carbon atoms, eight hydrogen atoms, and one oxygen atom. Most importantly, it is known as a heterocyclic ether. The term cyclic refers to compounds that have rings in their structure. The prefix ‘hetero’ tells us that at least one of the atoms in the ring is something other than carbon, and the ‘ether’ part of the name tells us the functional group. Ether is an organic functional group in which a central oxygen atom binds to two carbon-based groups. Another important structural aspect of THF is that it is a polar molecule. Because oxygen is electronegative relative to carbon and hydrogen, oxygen attracts electron density in the form of covalent bonds within the ring structure. As a result, as a polar molecule, THF is easily mixed with other polar species such as water, methanol, ethanol, and acetonitrile.
Production Process of Tetrahydrofuran:
- The most widely used industrial process for the production of THF involves the dehydration of 1,4- butanediol with an acid catalyst. This method is similar to the production of diethyl ether from ethanol. Butanediol is obtained from the concentration of acetylene with formaldehyde followed by hydrogenation.
- This method of producing tetrahydrofuran is the conversion of n-butane to maleic anhydride during the oxidation process and then catalytic dehydrogenation can produce tetrahydrofuran.
- Another method of production of diethylene oxide is catalytic dehydration of furan as an available compound.
THF Uses:
- The main use of tetrahydrofuran (THF) is as a solvent and it is used to produce many kinds of adhesives, cleaning and dyeing liquids, varnishes and printing inks, and many kinds of coatings.
- THF is a widely used solvent in the industry of PVC, natural and synthetic resins, vinyl films and cellophane, polymer coatings, protective coatings, and magnetic tapes. like Cyclohexanone
- This solvent is used in the pharmaceutical industry in the synthesis of drugs and vitamins and also in perfumery.
- This chemical is also used in the production of synthetic leather coatings.
- In many cases, the use of THF in the reaction allows higher efficiencies and faster reaction rates.
- as a solvent for polymers in gel permeation chromatography.
- The suitable solvent in reverse-phase liquid chromatography.
- Due to its good properties, solubility, and high penetration of THF solvent, it is used as a solvent for vinyl and epoxy adhesives.

- In addition, THF is used in pharmaceuticals, agriculture, and other industries as a reaction solvent and purifier.
Sell Tetrahydrofuran:
You can contact our experts at Shanghai Chemex for more information on THF pricing, and how to place an order.
Safety Information:
This solvent is almost a dangerous chemical and it is almost a highly flammable liquid. If inhaled, it will irritate the respiratory system and cause allergies. If THF solvent is absorbed into the skin, it can cause irritation. Contact of this organic compound with the eyes also causes eye irritation. Therefore workers must wear special work clothes, gloves, and masks When they are working with this organic substance because this substance is flammable.



First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and storage:
Keep the container tightly closed in a dry and well-ventilated place.






Reviews
There are no reviews yet.