Description
Cyclohexanone is one of the most widely used chemical solvents. It is found in both natural and synthetic forms and in small quantities is a major component of all petroleum products. However, due to its small amount, this material is produced industrially from catalytic hydrogenation of benzene. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
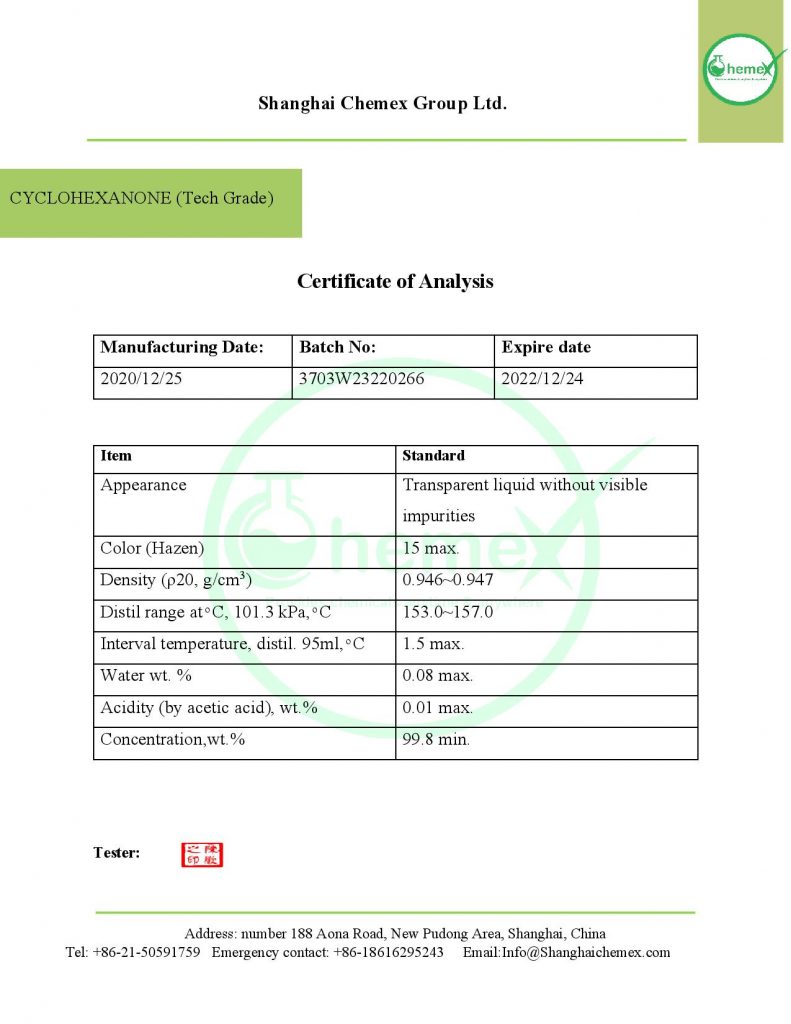
Cyclohexanone is a liquid and oily solvent with an odor similar to acetone and mint and has a yellow or colorless color. This solvent has a lower density than water, but its vapors are much heavier than air. Its main application is in the manufacture of nylon. This material is used as a chemical and solvent for the preparation and manufacture of nylon. It should be noted that this substance does not dissolve in water, but in acetone, ether, and benzene, it shows good solubility; The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | C6H10O |
| Molecular weight (g/mol) | 98.15 |
| Appearance | Colorless liquid |
| odor | peppermint or acetone-like |
| Density (g/cm3) | 0.9478 |
| Melting point (°C) | −47 |
| Boiling point (°C) | 155.65 |
| Color | white to pale-yellow |
| Form | Oily liquid |
| Chemical Structure Depiction | 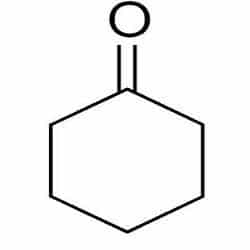 |
Structure and Formula of Cyclohexanone:
Cyclohexanone with the chemical formula C6H10O is composed of a six-carbon ring molecule attached to a ketone group. Which is found in two forms, boat, and chair.
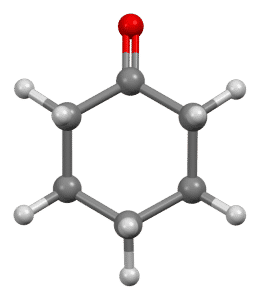
Cyclohexanone Production Process:
Commercial production of this material can be done using different methods:
- Hydrogenation of benzene is in a closed system
- It is prepared from the oxidation of cyclohexanol by chromic acid. In this process, chromic acid is obtained from a mixture of sodium bicarbonate or potassium bicarbonate and sulfuric acid.
- Hydrogenation of phenols:
C6H5OH + 2 H2 → (CH2)5CO
- Cyclohexanone is also prepared from the oxidation of cyclohexane in the air in the presence of a cobalt catalyst:
C6H12 + O2 → (CH2)5CO + H2O
- Catalytic dehydrogenation of cyclohexanol:
C6H11OH → C6H11OH + H2
- Cyclohexanone can be prepared from the oxidation of cyclohexanol with chromium trioxide called the Jones oxidation process.
Cyclohexanone As a Solvent:
Cyclohexanol is one of the best solvents in the industry and dissolves many chemical and organic compounds. for example:
- Ethanol and methanol
- Diethyl ether
- Xylene
- Diethylamine
- ethylene glycol
- Benzene and nitrobenzene
- Butyl acetate and ether
- As well as other organic solvents
Cyclohexanone Uses:
Cyclohexanone has various uses, the main and most important application of cyclo is its use in the nylon industry.
- As an intermediate in the production of nylon.
- Intermediate in the production of cellulose paints and varnishes.
- As a solvent in PVC printing ink. like Tetrahydrofuran

- As a solvent in herbicides , pesticides , plastics, and rubber
- In paint and varnish cleaners as a solvent.
- As a metal cleaner
- Solvent for cellulose acetate and nitrocellulose
- In rosy and silk staining
- Natural and synthetic resins
- As an active ingredient in antibacterial and antifungal compounds
- The main catalyst for making polyester and synthetic resins
Buy Cyclohexanone:
You can contact our experts in Shanghai Chemex to buy cyclohexanone and find out the price of this product through the communication channels available on the site.
Safety Information:
Cyclohexanone can cause significant damage. If inhaled, it can irritate the throat and respiratory tract, causing headaches and nausea. Excessive exposure of the eyes, nose, and skin to this substance and its vapors can cause similar effects to drowsiness and anesthesia. This substance belongs to the category of dangerous chemicals. This substance is highly flammable and its vapor explodes in the event of a spark. Therefore people who deal with this substance need to use special glasses, masks, and work clothes.


First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Cyclohexanone is a flammable chemical compound, so it should be stored in a tightly-closed container in a cool, dry, ventilated place away from heat or combustion sources.






Reviews
There are no reviews yet.