Description
Triethylene glycol is the third member of the two-factor alcohol series and is produced together with mono and diethylene glycol as a by-product of the direct hydration reaction of ethylene oxide. This material is used as a plasticizer in vinyl polymers and in the production of air disinfectants and other products. TEG is known for its ability to absorb high humidity and increase the dehydration of natural gas. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties of Triethylene Glycol:
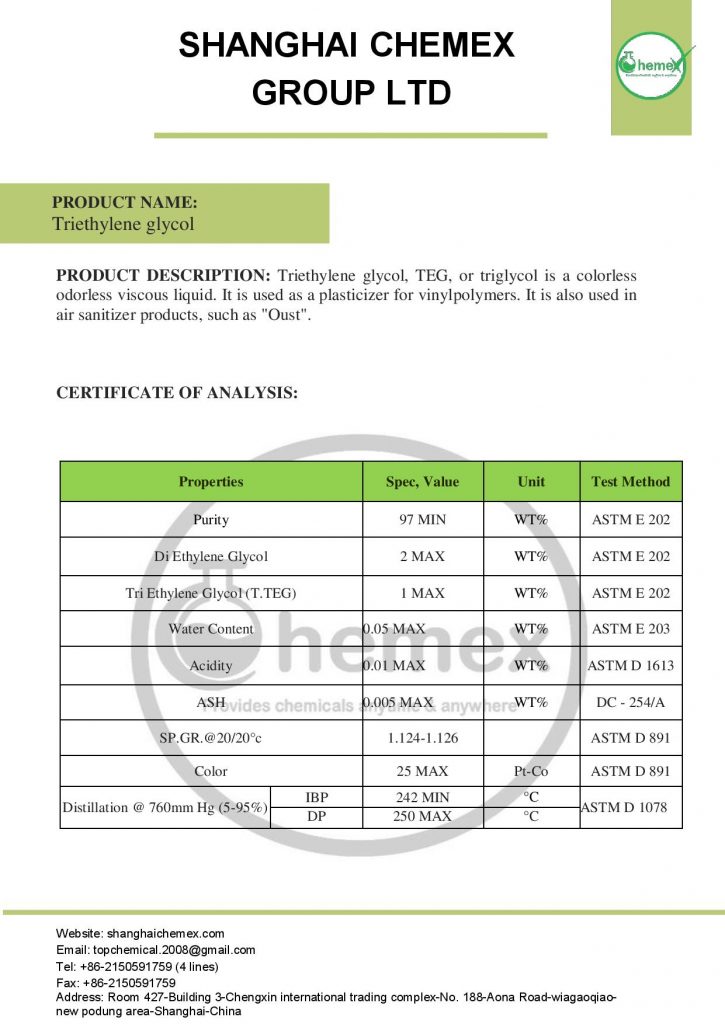
Triethylene glycol, also known as Tri glycol and TEG, is a colorless liquid with high viscosity and boiling point, and low volatility. This chemical is easily soluble in water and its boiling point is more than mono ethylene glycol and diethylene glycol. In terms of solubility, it can be said that it is completely compatible with water and it is completely soluble in it. It is also completely soluble in most solvents such as ethanol or acetic acid, glycerin, pyridine, and also aldehydes. Ethylene glycol is very similar to glycerin in terms of physical and chemical properties and can be used as a glycerin substitute. The most important physical and chemical properties of this compound can be summarized in the following table:
| Name | Triethylene glycol |
| Chemical formula |
C6H14O4 |
| Molecular weight (g/Mol) | 150.17 |
| Appearance |
Colorless liquid |
| Odor | Practically odorless |
| Density (g/cm³) | 1.1 |
| Melting point (° C) | -7.0 |
| Boiling point (°C) | 285 |
| Flash Point (°C) | 165 |
| Chemical Structure Depiction |
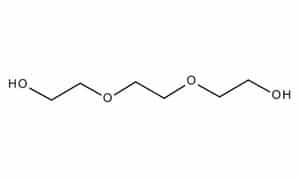 |
Production Process of TEG:
In the industrial method of production of this alcohol, which we will discuss below, the high purity of this material can be produced. In this method, pure ethylene oxide or a mixture of water and ethylene oxide is heated to prepare for the reaction. During the thermal process, ethylene oxide is converted to mono ethylene glycol (MEG), diethyl glycol (DEG), and triethylene glycol (TEG) is produced as a by-product in small quantities. To achieve high purity mono ethylene glycol (MEG), excess water needs to be evaporated and used in a few steps, during which the evaporators produce low-pressure heaters.
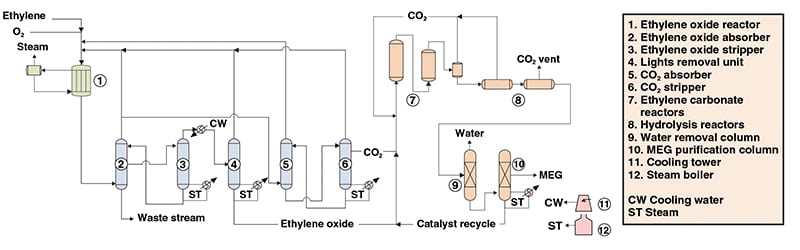
Raw glycol mixtures are rapidly classified into successive vacuum columns using catalysts. Numerous factors influence the purity of this material, such as the order of feeding. Most ethylene glycol production units are adjacent to ethylene oxide production units, but there are also separate ethylene glycol production units. This method is one of the most famous methods of production of TEG in the world and the quality of the product produced by it is very high.
Triethylene Glycol Uses:
Oil and Gas Industry:
Triethylene glycol is used in the oil and gas industry to dehumidify natural gas. It may also be used to dehydrate other gases, including CO2, H2S, and other oxygenated gases. Natural gas needs to dry to a certain point because the moisture in natural gas can freeze pipelines and create other problems for end-users of natural gas.
Disinfectant Manufacturing Industries:
Triethylene glycol is known as a relatively mild disinfectant for a variety of bacteria, influenza A viruses, and spores of the fungus Penicillium notatum. However, the extremely low toxicity, wide compatibility of the material, and low odor along with its antimicrobial properties indicate that it is ideal for air disinfection purposes in areas with the possibility of pollution.
Iron Rust:
To prevent iron oxidation in the long run, resin coatings or stainless steel and insulation based on phenolic resins and epoxies are used. Because triethylene glycol is widely used in the manufacture of these materials, it can be considered an anti-corrosion agent for iron and inhibits its oxidation.

Textile Industry:
Triethylene Glycol is used in the textile industry as an air conditioning and lubricant in the production of a large number of natural and synthetic fibers.
Resins:
Triethylene glycol is mainly used in the production of unsaturated polyester resins.
Buy Triethylene Glycol (TEG):
For more information on purchasing and ordering this product, please contact our experts in Shanghai Chemex through the numbers on the site.
Safety Information:
This compound is in the category of toxic substances, so be sure to follow the safety tips when working with it.
In case of contact with the eyes, may cause redness, burning, and watering. It is also very dangerous to swallow and causes severe gastrointestinal poisoning. Inhalation may also lead to confusion and nausea.
First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person into the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
This chemical is non-volatile and corrosive and should be in closed containers in a dry and cool place away from heat and direct sunlight.






Reviews
There are no reviews yet.