Description
Phosphorus pentoxide is an anhydrous (anhydrous) form of phosphoric acid. Its chemical formula is P2O5, but since this compound is unstable, it forms P4O10.

Phosphorus pentoxide is used as a dehydrating agent in organic synthesis, which is used to convert primary amides to nitriles and mineral acids to their anhydrides. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.
Physical and Chemical Properties:
Phosphorus pentoxide appears as white crystals. This compound is highly hygroscopic, meaning it has a strong attraction to water, and its reaction with water is highly calorific.
The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | P2O5 |
| Molecular Weight (g/mol) | 141.94 |
| Appearance | Solid |
| Density (g/cm3) | 2.39 |
| odor | odorless |
|
Melting point (° C) |
340 |
| Boiling point (° C) | 360 |
| Solubility | soluble in water and sulfuric acid
Insoluble in acetone and ammonia |
| Color | White |
| Form | Powdery or crystalline |
| Other names | Diphosphorus pentoxide
Phosphorus(V) oxide Phosphoric anhydride |
| Chemical Structure Depiction | 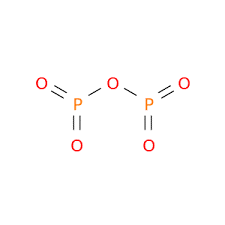 |
Formula and Structure of Phosphorus Pentoxide:
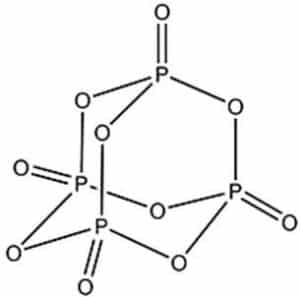
Phosphorus pentoxide can be found in countless crystal structures (polymorphs) but the most common and stable structure found is the one shown in the image below:
Phosphorus Pentoxide Production Process:
Anhydrous phosphorus pentoxide is produced by burning yellow phosphorus in dry air. P2O5 (or P4O10) can be prepared by burning tetra phosphorus in an environment containing sufficient oxygen. The chemical reaction for the preparation of P2O5 is presented below:
P4 + 5O2 → 2P2O5
This compound is a source of pure phosphoric acid.
Reaction with Water:
Reacts with cold water to produce toxic metaphosphates:
P2O5 + H2O→2HPO3
In Reacts to hot water, it produces toxic orthophosphate:
P2O5 + 3H2O→2H3PO4
Phosphorus Pentoxide Uses:
- As a strong desiccant and disinfectant
- Many reactions that require the removal of a water molecule can use P2O5, such as dehydration of amide to nitrile.
- Phosphorus pentoxide Raw material for phosphate esters as surfactants and extractants
- This compound can be used to manufacture special catalysts for phosphoric acid.
- Used as a Condensing agent in organic synthesis
- It is also used in the manufacture of optical glass.
P2O5 Fertilizer:
This compound can be used as an agrochemical to produce plant fertilizers. Phosphorus is the main element of plant growth.
This fertilizer is suitable for all crops and all soils. In acidic soils, it should be used with organic fertilizers. like monopotassium phosphate

Buy Phosphorus Pentoxide:
Shanghai Chemex, active in the field of chemical sales, offers phosphorus pentoxide with the best quality and the most appropriate price to customers. To buy this product, please contact our experts through the communication channels available on the site.
Safety Information:
- The pack causes damage if swallowed.
- In case of contact with skin, it causes inflammation.
- It will cause severe inflammation and irritation of the eyes.
- If inhaled, it can cause allergies in the respiratory tract.

First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Store phosphorus pentoxide in a tightly closed container in a cool, dry, well-ventilated place away from incompatible materials.






Reviews
There are no reviews yet.