Description
Sodium hexametaphosphate (SHMP) with the chemical formula Na6P6O18 is a commercial polymer mixture of metaphosphates and is also known as Calgon or Graham Salt, this product is used as an emulsifier with E452i index in most food products. This compound is one of the most widely used materials in water purification and food production industries. SHMP is in the category of Industrial chemicals of Shanghai Chemex group Ltd.

Physical and Chemical Properties:
Sodium hexametaphosphate appears as white crystals that are soluble in water. This compound has the ability to absorb air moisture and can react with metal ions. It is used as a receptor and is used in a wide range of industries, including food additives. In the table below some of the chemical and physical properties of this product are mentioned:
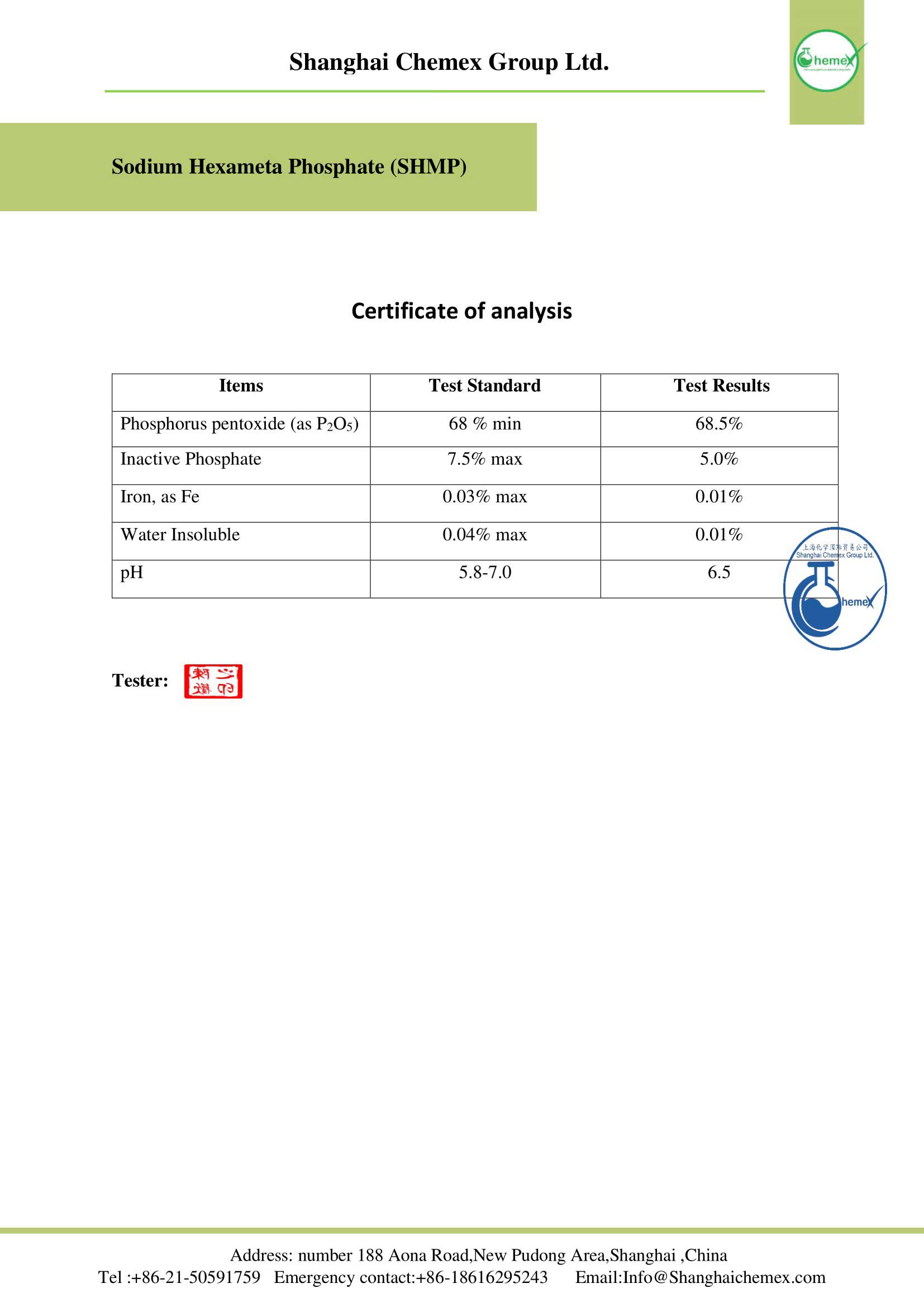
| Chemical formula | Na6P6O18 |
| Molar mass (g/mol) | 611.7704 |
| Appearance | solid |
| Odor | odorless |
| Density (g/cm3) | 2.484 |
| Melting Point (°C) | 628 |
| Boiling point (°C) | 1,500 |
| Solubility in water | soluble |
| Solubility | insoluble in organic solvents |
| Color | white |
| Form | crystals |
| Synonyms | Calgon S, Glassy sodium, Graham’s salt,
Hexasodium metaphosphate, Metaphosphoric acid, hexasodium salt |
| Chemical Structure Depiction | 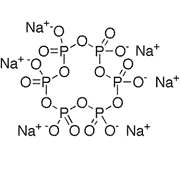 |
The Structure of Sodium Hexametaphosphate:
The chemical composition of sodium hexametaphosphate, in fact, is a similar structure to polymers, which are formed by metaphosphate chains in a hexagonal structure. In the molecular structure of sodium hexametaphosphate, six sodium cations and six phosphates are linked together as a polymer chain.
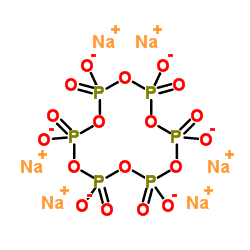
Production of Sodium Hexametaphosphate:
To produce this material, a two-step reaction using ortho monosodium phosphate and monosodium pyrophosphate is used and heat is added according to the following reaction equation.
The second step in the synthesis of this product is heating with pyrophosphate. The two-step reaction equation for the production of this product is as follows:
- 2NaH2PO4 → Na2H2P2O7 + H2O
- 3Na2H2P2O7 → (NaPO3)6 + 3H2O
This is followed by rapid cooling.
Sodium Hexametaphosphate Uses:
The product has a wide range of applications in various industries such as industrial applications, paper making, dyeing, textile, petrochemical industry, water cooling and hardening, metallurgy, and construction to use in the food industry as food additives such as syrups, cheese powder, fish fillets, salad dressing, and beer.
- Use as additives in the food industry (adding this compound to food preserves better color, taste, and flavor in foods).
- Used as a water softener to produce detergents. like Propylene carbonate
- Adjust the pH of the solution to 8-8.5.
- As a particle suspender (this property is used for suspension and colloid solutions in various industries).
- Use as a toothpaste activator.
- Additive to raw materials of the pharmaceutical industry.
- Removal of copper and zinc ions in the water treatment industry.
- As a sediment inhibitor in reverse osmosis systems.
- Used as an anti-corrosion agent to prevent rust pipes.
- Formation of a complex to remove disturbing substances in water
- Used in photography to prevent stains on film and photos.
What is Sodium Hexametaphosphate used for in Water Treatment?
Divalent and trivalent cations are used in aqueous solutions to coagulate insoluble suspension particles, especially negatively charged colloidal particles. Therefore, hexametaphosphate is used for water treatment. Sodium carbonate is sometimes added to the above product to increase its acidity to 8.6 to be used as a detergent and as a dispersing agent and homogenizer to reduce water hardness. This material prevents the accumulation of suspended particles on a large scale to speed up their settling process.

How is SHMP used in Food?
It is used in combination with other emulsifying salts in the production of processed cheeses. These products change the pH and prevent spoilage of food, stabilize natural food pigments and maintain the radiance and quality of these materials.

Buy Sodium Hexametaphosphate:
Shanghai Chemex Group Ltd, as a manufacturer, supplier, and seller of chemical raw materials in China, is ready to provide any services in the field of chemical sales to our valued customers. This product is sold with quality assurance in accordance with the analysis provided. For more information on the price of sodium hexametaphosphate, and how to place an order, contact our experts.
Safety Information:
This substance contains phosphorus. Phosphorus is a known nutrient for humans. But when consumed in excess, it can upset the balance of other elements in the body and negatively affect the kidneys, heart, and bones. Complications that can result from the overuse of this substance are hyperplasia, osteoporosis, severe skin irritations, etc. Also, due to the presence of sodium in this substance, its consumption should be limited to people who have a salty diet and avoid sodium.

First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Store in a tightly-closed container in a cool, dry, well-ventilated place.






Reviews
There are no reviews yet.