Description
Vinyl sulfonate is an organosulfur compound with the formula CH₂ = CHSO₃H. It is one of the simplest unsaturated sulfonic acids. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.
physical and chemical properties:
Vinyl sulfonate is a colorless, water-soluble compound, but commercial specimens may appear yellow or red; The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | C2H4O3S |
| Molar mass(g/mol) | 108.11 |
| Appearance | Colorless liquid |
| Boiling point(° C) | 128 |
| PH | 8-12 |
| Solubility | soluble in water |
| Color | Light yellow |
| Form | liquid |
| Chemical Structure Depiction | 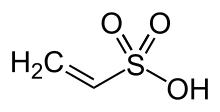 |
Vinyl sulfonate production process:
- Produced in the industry through alkaline hydrolysis of carbonyl sulfate and acidification of sodium vinyl sulfonate:
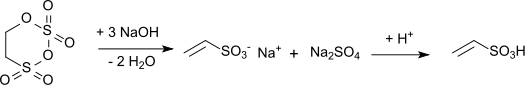
- This compound is also produced by dehydrating ethionic acid with phosphorus pentoxide:
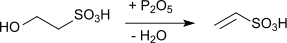
Vinyl Sulfonate Uses:
- Vinyl sulfonate monomer is used in the preparation of homopolymers and highly acidic or anionic copolymers.
- Used as a brightener and leveler in nickel baths.
- also as an intermediary in the pharmaceutical industry, Surfactants, and organic synthesis are used.
- It is used as a photoresist in the electronics industry, and as an ion-conducting polymer electrolyte membrane for fuel cells.
Safety Information:
Irritant to eyes, skin, mucous membranes, and respiratory system. May be harmful by ingestion, skin absorption, and inhalation.
First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Store in a cool, dry, well-ventilated area away from heat and sources of ignition.


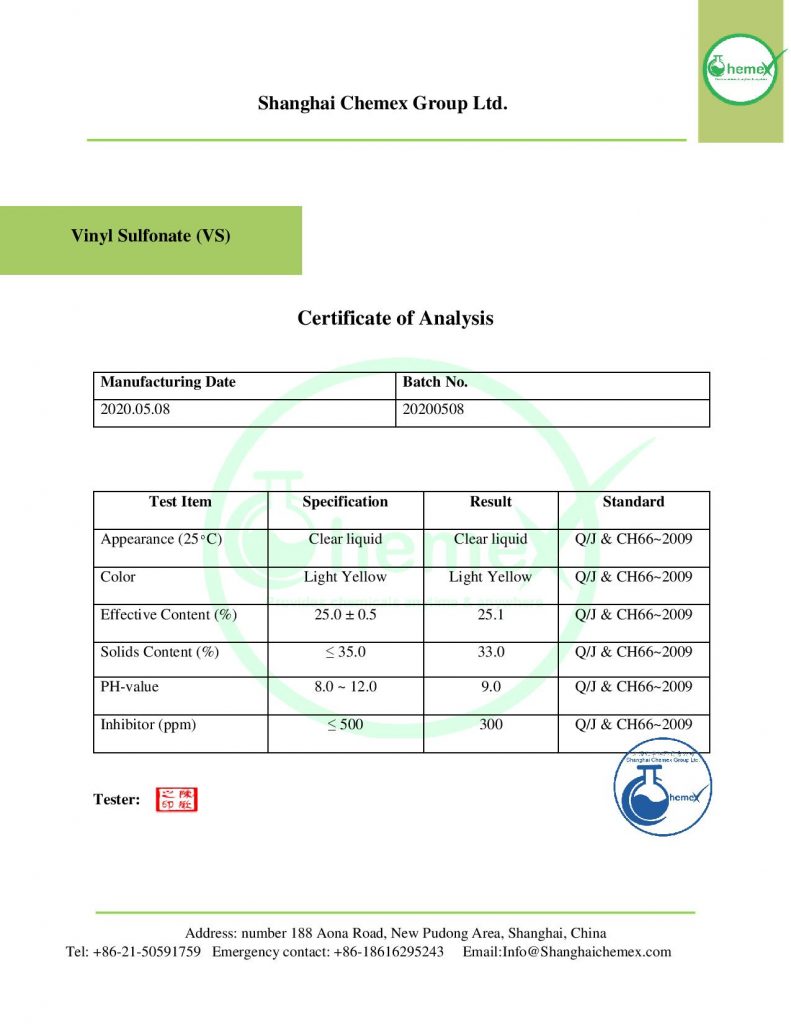




Reviews
There are no reviews yet.