Description
Zinc stearate is formed by the reaction of zinc sulfate solution and sodium stearate. This compound has different ratios of palmitic and stearic acids. Zinc stearate can be used as a preservative or water repellent in ointments and powders to treat acne and other skin conditions. It is also used as a lubricant to improve the texture of cosmetic products and has antiseptic and topical properties; Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
Zinc stearate is a chemical compound with a soft, white powder appearance. The compound is insoluble in water, alcohol, and ether, but dissolves in aromatic solvents such as benzene and chlorinated hydrocarbons with increasing heat. It is a hydrophilic and lipophilic compound and is produced in the form of flakes, granules, and powder.
| Chemical formula | C36H70O4Zn |
| Molecular Weight (g/Mol) | 632.3 |
| Appearance | Solid |
| odor | Faint odor |
| Density (g/cm3) | 1.095 |
| Melting point (° C) | 120 to 130 (248 to 266 °F; 393 to 403 K) |
| Water Solubility | insoluble |
| Solubility | Soluble in acids and aromatic solvents when hot |
| Flash Point (°F) | 530 |
| Vapor Pressure (mm Hg at 25 °C) | 0 |
| Synonyms | Zinc octadecenoate, Zinc distearate, Dermarone |
| Color | White |
| Form | Fine, soft, bulky powder |
| Chemical Structure Depiction | 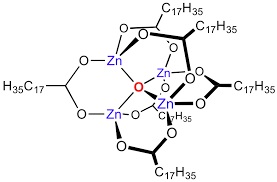 |
Production Process of Zinc stearate:
The industrial production process of this product is two-step:
The first step involves the neutralization process and reaction. In this step, stearic acid is neutralized by reaction with sodium hydroxide, then in the second step, zinc sulfate is used for the final production of the product. One method of producing ZnSt2 is by using sodium stearate and zinc sulfate in a Sedimentation process, and stearic acid and zinc oxide in a fusion process according to the purity of the product.
Zinc stearate Uses:
The three main uses of zinc stearate: as a heat stabilizer, Releasing substance, and Lubricant.
- The most important application of this chemical is as a lubricant in the rubber industry.
- It is used as a lubricant, release agent, and halogen absorber in the paint and plastics industries.
- Used in the formulation of cosmetics and personal care products such as eyeliner, eye shadow, mascara, lipstick, blush, powder cream, and cream.
- Application in the paper industry.
- Used in the concrete industry.
- Zinc stearate is used as an activator in welding rubber.
- Used in the concrete industry.
- Other uses include washing powder, textiles, building materials, inks, and wax compounds.
Zinc stearate in rubber processing:
The main application of this material as a raw material in the rubber industries. In the past, stearic acid and zinc oxide were commonly used in the rubber heating process. These materials are actually a kind of activator and accelerator of the process. Based on this, it can be expected that zinc stearate can also play an important and fundamental role in the rubber heating process.

How is zinc stearate used in plastic?:
- This material is used as a lubricant and stabilizer in the production of PVC.
- It is used as a pigment dispersion in master batches.
- Due to the transparency of this product, it is used in the production of glass polystyrene and other transparent polymers.
- Used as a metal release agent in polyurethane.

Safety information:
- Inhalation: May experience dizziness and headache. Irritation of the nose and throat.
- Ingestion: May cause gastrointestinal irritation, nausea, and vomiting.
- Skin contact: Mild irritation of the skin may occur.
- Eye Contact: Mild irritation of the eye tissue may occur


First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person into the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and storage:
Keep in a tightly closed container, stored in a cool, dry, ventilated area. Protect against physical damage.






Reviews
There are no reviews yet.