Description
chrome oxide is an inorganic compound with the chemical formula Cr2O3, which is a combination of oxygen and chromium and is known as green chromium. This oxid occurs naturally and is a highly insoluble compound with high thermal stability and acid-base properties. Due to its excellent stability, this material is used as a green pigment in inks, glass, and paints, as well as as a coloring agent in the production of ceramics and to create a matte green color in glazes. Shanghai Chemex is one of the most reputable manufacturers of this chemical in the world.

Physical and Chemical Properties of Chrome Oxide:
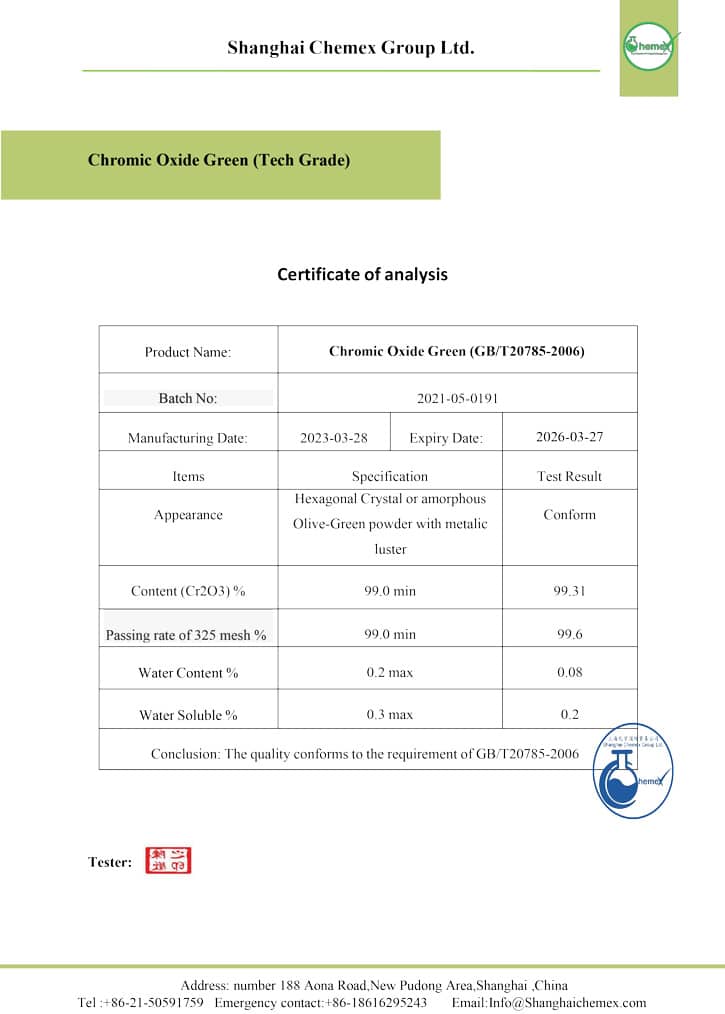
This inorganic compound is in the form of green crystal powder, which has high strength and is resistant to heat and sunlight. It is also insoluble in water and difficult to dissolve in acids; The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | Cr2O3 |
| Molecular Weight (g/mol) | 151.9904 |
| Appearance | Light to dark green, powder |
| PH | 5-9 |
| Odor | Odorless |
| Density (g/cm3) | 5.22 |
| Melting point (° C) | 2,435 |
| Boiling point (° C) | 4,000 |
| Solubility in water | insoluble |
| Solubility | insoluble in alcohol, acetone, acids |
| Color | Light to dark green |
| Form | crystals |
| Chemical Structure Depiction | 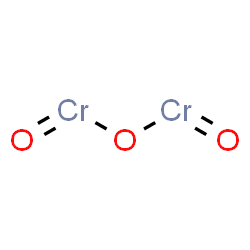 |
The Chemical Formula for Cr2O3:
Chrome oxide is an ionic compound, this chemical must be electrically neutral, meaning that the positive charge of the cation is balanced by the negative charge of the anion.
2×[Cr]3++3×[O]2−→Cr2O3
Chrome Oxide Nanoparticles:
Nano-scale chrome oxide particles are naturally between 10 and 30 nanometers, and their specific surface area is between 130 and 150 square meters per gram. These nanoparticles are used sparsely in nanofluids. Nanofluids are nanoparticles that are formed in a solution in the presence of surfactants.
Production of Chrome Oxide:
- In the reaction between sodium dichromate and sulfur at very high temperatures, Chrome oxide is obtained.
Na2Cr2O7 + S → Na2SO4 + Cr2O3
- This oxide compound is also obtained from the decomposition reaction of ammonium dichromate, which is an exothermic reaction.
(NH4)2Cr2O7 → Cr2O3 + N2 + 4 H2O
Chrome Oxide Uses:
- In addition to its use as paint in ceramics, chrome oxide is also used in the paint and ink industry, as well as in the glass industry. like cobalt oxide

- Chrome oxide coatings or black oxide coatings are industrial coatings that are very effective in sealing surfaces as well as abrasion resistance. These coatings have high chemical resistance and are also anti-reflective.
- This powder composition can be mixed with clay and used in metal glazes.
- This green pigment is used in cosmetic formulations. This combination is approved for cosmetic use such as hair color, nails, and cleansers, but its use is not allowed for lip makeup.
- This green crystalline powder is also used as a catalyst.
- Chrome oxide can produce colors such as red, brown, yellow, and especially green.
Chrome Oxide Green Pigment:
Chrome oxide green pigment is widely used in tiling, polishing, painting, plastics, and construction materials due to its excellent performance in green, erosion resistance, corrosion resistance, and chemical resistance.
Buy Chrome Oxide:
Shanghai Chemex is currently one of the suppliers and manufacturers of chemical products in China, which provides a wide range of chemicals to its customers. To place an order and purchase this product, contact our experts.
Safety Information:
Inhalation of this green powder stimulates the respiratory system. Prolonged contact with the skin causes ulceration and allergies. The dust of this powder irritates the eyes and Also, and this compound is not flammable. If ingested can cause nausea and other problems.
Packing and Storage:
Keep the container tightly closed in a cool, well-ventilated area and protected from physical damage.







Reviews
There are no reviews yet.