Description
In general, fumed silica consists of microscopic droplets of amorphous silica and is a pure and relatively reactive source of silica. This material is a very light and fluffy powder that has a very low apparent density.
It can be produced in a wide range of sizes and surfaces as hydrophilic and hydrophobic. This compound is somewhat more expensive than other sources of silica and is used as a thickening, drying, anti-sticking and stabilizing agent in medicines, cosmetics, paints and coatings, sealants and batteries.
Properties:
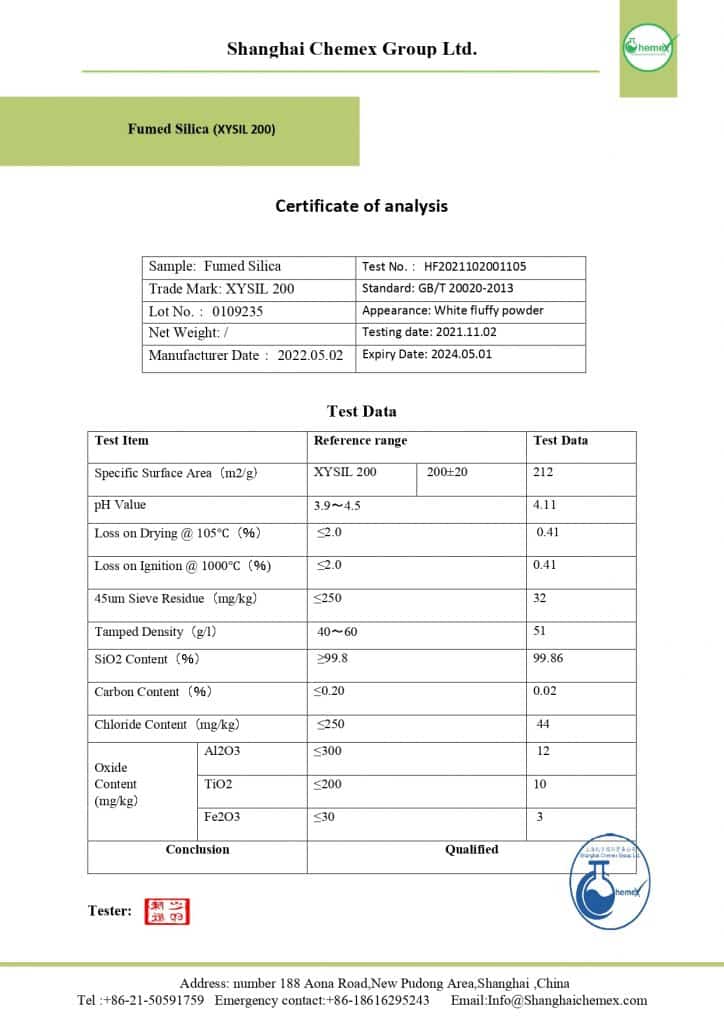
Fumed silica has a primary particle size of 5 to 50 nm and non-porous particles. This material has a very strong thickening effect and has a density of 160-190 kg/m3. In the table below, you can see some of the physical and chemical properties of this substance.
| chemical formula | SiO2 |
| Molecular Weight | 60.08 g |
| Appearance | White powder |
| Melting Point | 1,600° C |
| Boiling Point | 2,230° C |
| Density | ~2.3-4.5 g/cm3 |
Production:
Fumed silica is synthesized by the pyrolysis method, in which silicon tetrachloride is made either from quartz sand vaporized with oxygen or hydrogen in an electric arc at a temperature of 3000 degrees Celsius.
This process is carried out in the liquid phase and silica nanoparticles are synthesized with an acidic or basic catalyst and an alcoholic solvent in the presence of silicon alkoxide.
Applications of silica fumed:
This material has many applications in the industry, including:
- Thickening agent in powders
- Like silica gel as a desiccant
- In cosmetics
- As an abrasive in toothpaste
Also, other uses of this material include filling in silicone elastomer and adjusting viscosity in paints, coatings, printing inks, adhesives and unsaturated resins.
Safety:
Fumed silica is not known as a carcinogen. It has a fineness and thinness that can easily be carried in the air and its inhalation causes irritation.







Reviews
There are no reviews yet.