Description
Potassium sulfate contains two essential elements potassium and sulfur for plant growth, which are used as fertilizer in the agricultural industry. Other applications of this product include various industries such as chemical, pharmaceutical, and food industries. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
Potassium sulfate with the chemical formula K2SO4 usually appears in the form of white crystals with a salty and bitter taste, which is soluble in water but insoluble in ethanol; In the table below some of the chemical and physical properties of this product are mentioned:
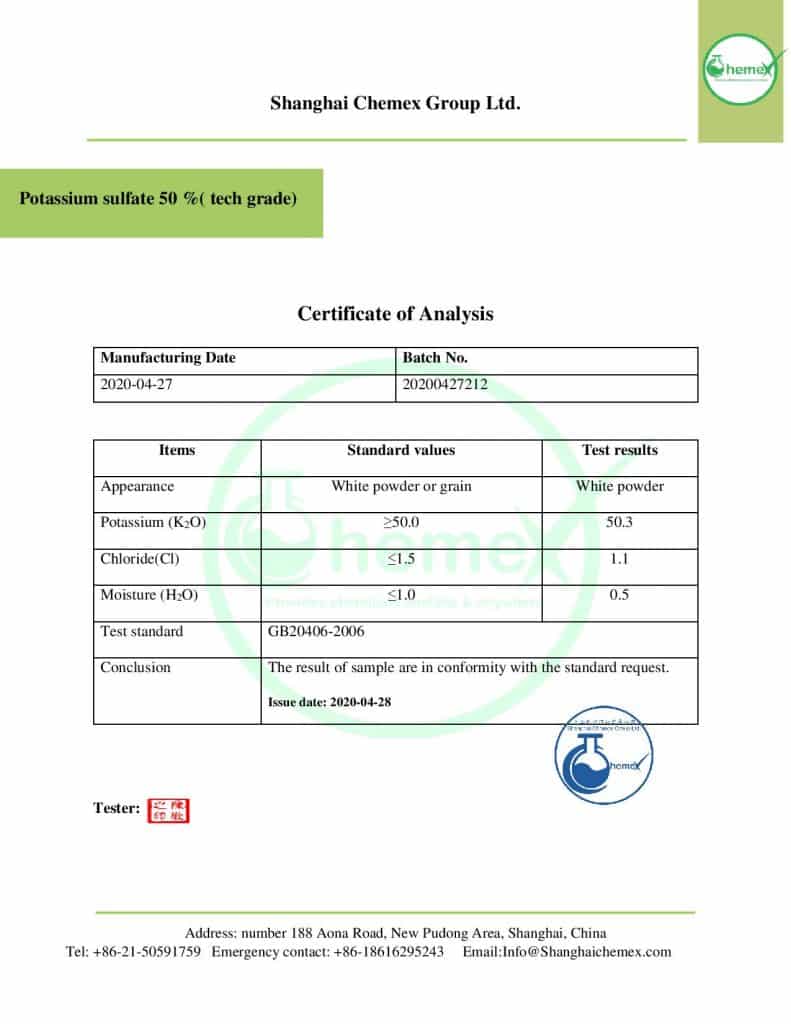
| Chemical formula | K2SO4 |
| Molar mass (g/mol) | 174.26 |
| Appearance | solid |
| PH (5 % solution) | Between 5,5 and 8,5 |
| Odor | Odorless |
| Taste | Hard, bitter, saline taste |
| Density (g/cm3) | 2.66 |
| Melting Point (° C) | 1067 |
| Boiling point (° C) | 1689 |
| Solubility in water | Freely soluble in water |
| Solubility | insoluble in ethanol, acetone, carbon disulfide |
| Color | Colorless or white |
| Form | crystals or crystalline powder |
| Synonyms | Potassium sulfate, Dipotassium sulfate,
Sulfuric acid dipotassium salt |
| Chemical Structure Depiction | 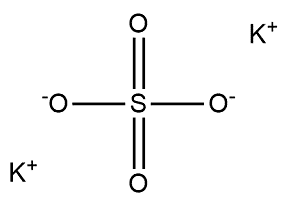 |
Potassium Sulfate Production Process:
The production of this chemical is done in different ways: The reaction of potassium chloride with Langbeinite ore, the reaction of potassium chloride with sulfuric acid or sulfur dioxide, and also the recycling of sugar waste are some of the methods used to produce this chemical compound.
Potassium Sulfate Uses:
This known chemical compound has many applications in industry such as Agrochemicals (non-pesticides), Catalysts, Laboratory chemicals
- The most important use of potassium sulfate in agriculture is to meet the plant’s need for potassium and sulfur.
- Potassium sulfate reduces soil acidity and helps to better absorb iron and phosphorus.
- Strengthens plant nodes with high carbohydrates such as potatoes.
- Improve the color quality and taste of fruits.
- In the food industry as a flavoring.
- As a substitute for common salt (sodium chloride).
- This compound is also used in the glass industry.
What is Potassium Sulfate used for in Agriculture?
Most of the consumption of potassium sulfate is in the agricultural industry and as an agricultural fertilizer to supply potassium and soil sulfur. Potassium is needed to complete many essential functions in plants, including activating enzymatic reactions, synthesizing proteins, forming starches and sugars, and regulating water flow in cells and leaves. Often the concentration of K in the soil is too low to support the growth of a healthy plant.
One of the most important and best properties of potassium sulfate should be noted that, unlike other fertilizers, it does not cause soil salinity, so it is very suitable for products that are sensitive to chlorine. These agricultural products include pistachios, tea, citrus fruits, tobacco, potatoes, berries, grapes, etc. Of course, it should be noted that chemical fertilizers such as potassium sulfate are not suitable for nitrogen-based products and do not have the necessary efficiency in these cases.

Buy Potassium Sulfate:
Shanghai Chemex is currently one of the suppliers of chemical products in China, which sells potassium sulfate in agricultural grade with the best price and quality. Contact our experts to buy the product and place an order.
Safety Information:
- Inhalation of this sulfate causes coughing or sneezing.
- Its oral consumption in high amounts causes disorders and irritation in the digestive system.
- In contact with the skin and eyes, it does not pose a problem for human health, but the entry of dust into the eye causes mechanical irritation in the eye.
First-aid measures:
- Eye Contact: In case of contact with the eyes, immediately flush the eyes with plenty of water.
- Skin Contact: Remove this material from the skin with plenty of soap and water.
- Inhalation: Remove to fresh air immediately. If not breathing, give artificial respiration.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Store in a cool, dry, well-ventilated area away from incompatible substances and direct sunlight.






lina –
Will potassium sulfate lower pH?
china chemicals –
Potassium sulfate cannot be used to lower soil pH, but this fertilizer causes confusion on this issue because it contains sulfur and because it sounds similar to aluminum sulfate; both of these materials are used to acidify the soil.