Description
Sodium bromide is a white powder with the chemical formula NaBr, which is a non-mineral chemical compound. The substance with square crystals is one of the main sources of bromide ion production and this compound can be prepared by the reaction of sodium hydroxide with hydrogen bromide. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
Sodium bromide is a mineral compound that appears in the form of a white crystalline powder with a salty and somewhat bitter taste with a smell similar to sulfur dioxide. This compound is soluble in water, alcohol, and liquid ammonia and insoluble in acetone; The most important physical and chemical properties of this compound can be summarized in the following table:
| Chemical formula | NaBr |
| Molecular Weight(g/mol) | 102.894 |
| Appearance | Solid |
| odor | the odor of sulfur dioxide |
| Taste | Saline |
| Density (g/cm3) | 3.21 (anhydrous)
2.18 (dihydrate) |
| PH | 6.5-8.0 |
| Melting point (° C) | 747 (anhydrous)
36 (dihydrate) |
| Boiling point (° C) | 1,390 |
| Solubility | Soluble in water, alcohol, liquid ammonia, pyridine, hydrazine, SO2, amine
Insoluble in acetone, acetonitrile |
| Color | White |
| Form | crystals, granules, or powder |
| Chemical Structure Depiction | 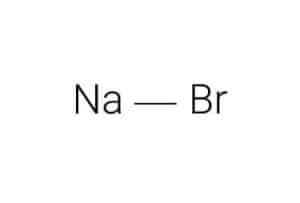 |
Structure of Sodium Bromide:
Sodium is a positively charged radical that is highly volatile. The element reacts with the negatively charged bromide radical to form a predominantly stable ionic bond that forms the sodium bromide compound.

Sodium Bromide Production Process:
Sodium bromide can be prepared in several ways:
- From the reaction of sodium hydroxide with hydrogen bromide:
HBr + NaOH → NaBr + H2O
- From the reaction between the bromine solution and sodium hydroxide:
6NaOH + 3Br2 → 5NaBr + NaBrO3 + 3H2O
Sodium Bromide Uses:
- In medicine as an anticoagulant, anticonvulsant and sedative; like Trisodium citrate
- The main consumer of sodium bromide in the oil and gas drilling industry
- As a disinfectant, detergent, and as a reagent in the pharmaceutical industry
- As a catalyst in oxidation reactions
- In the preparation of liquid photography film as a raw material
- As a bromine agent in the printing and dyeing industry
- The perfume industry produces synthetic perfumes

Buy Sodium Bromide:
To buy Sodium bromide and find out the price of this product through the communication channels available on the site, you can contact our experts in Shanghai Chemex.
Safety Information of Sodium Bromide:
- Causes mild eye irritation
- It May cause skin irritation. May be harmful if absorbed through the skin.
- May cause irritation of the digestive tract. Symptoms may include headache, excitement, fatigue, nausea, vomiting, stupor, and coma. May be harmful if swallowed.
- Inhalation of high concentrations may cause central nervous system effects characterized by nausea, headache, dizziness, unconsciousness, and coma. May cause respiratory tract irritation.


First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately.
Packing and storage:
It is better To store it in a cool and dry place. Store in a tightly-closed container and protect it from moisture.






Reviews
There are no reviews yet.