Description
Benzotriazole, also known as BTA, is a chemical used as a ligand to form complexes and has many applications in industry, the most common of which is to prevent rust and corrosion of metals. This chemical is a corrosion inhibitor for metals such as copper and its alloys, silver, aluminum, cobalt, and zinc, which are relatively soluble in water and do not decompose easily. Shanghai Chemex is one of the most reputable suppliers of this chemical in the world.

Physical and Chemical Properties:
The appearance of Benzotriazole is in the form of white burning crystals. It is soluble in alcohol, chloroform, and benzene, toluene, and dimethylformamide. This compound is relatively insoluble in water and can not be easily decomposed. Due to its strong chemical and thermal stability as well as hydrophobicity, it is used in water treatment and drug production.
In the table below some of the chemical and physical properties of this product are mentioned:
| Chemical formula | C6H5N3 |
| Molecular Weight (g/mol) | 119.127 |
| Appearance | Solid |
| odor | odorless |
| Density (g/cm3) | 1.36 |
| Melting point (° C) | 100 |
| Boiling point (° C) | 350 |
| Solubility | Soluble in water, ethanol, benzene, chloroform, toluene, DMF |
| PH | 5 – 6.5 |
| Color | white to light tan |
| Form | crystalline powder |
| Chemical Structure Depiction | 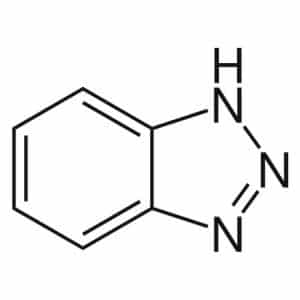 |
Structure and Formula of Benzotriazole:
This substance is a heterocycle compound with the chemical formula C6H5N3. Benzotriazole has two nitrogen rings in its chemical structure which are produced by the fusion of the benzene ring, this point causes the stability and resistance of the molecule.
This compound consists of two five-membered rings that include tautomers A and B, and derivatives of both tautomers can form structures C and D.
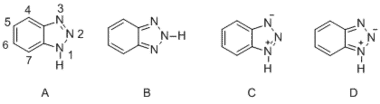
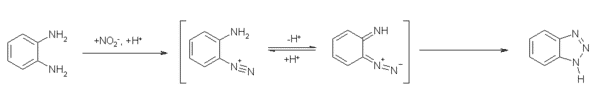
Benzotriazole Production Process:
Benzotriazole is synthesized from the reaction between orthophenylenediamine, sodium nitrite, and acetic acid. To improve the reaction, it is done at temperatures of 5 to 10 degrees and in an ultrasonic bath. The purity percentage of the product is slightly higher than 98.5%. The general description of this process is as follows:
Benzotriazole Uses:
- Benzotriazole is a very widely used chemical in various industries.
- As a corrosion inhibitor for metals such as copper and its alloys
- Anti blur factor in photo printing
- Production of light-sensitive chemicals
- Application in cleaners used in plating processes
- Production Water purification products and Laboratory chemicals
Benzotriazole Corrosion Inhibitor:
One of the most common methods of corrosion control is the use of disinfectants such as nitrates and chromates. Finding alternative compounds for these compounds is very important for environmental reasons. Benzoazole and the azole family are one of these alternatives, which chemically adsorb nitrogen atoms on the surface by sharing unpaired electron pairs with an empty metal orbital to prevent dissolution and corrosion. like Morpholine
This product consists of benzene and a tri-azole ring. The nitrogen atoms in the triazole ring form a bond between the copper, which inhibits the product.

Application of Medicinal Benzotriazole:
Heterocyclic compounds make up a wide range of drugs and are used to treat many diseases. One of the most important heterocyclic compounds is triazole derivatives. Triazole analogues such as imidazole, thiazole, carbazole, oxazole and benzotriazole are among the most important drug combinations. In recent years, researchers have conducted extensive research into the effects of benzotriazole on cancer.

Buy Benzotriazole:
Shanghai Chemex, as a supplier of chemical products, provides customers with a wide range of chemicals used in various industries. Contact our experts to buy Benzotriazole and also to know the selling price of this substance.
Safety Information:
- Inhalation of this substance can cause respiratory distress.
- Avoid eating it as swallowing it can cause serious harm.
- Using this substance can cause itchy skin and eyes. As a result, the use of glasses, masks, and gloves is always recommended.
- By heating this substance, toxic compounds such as carbon dioxide and nitrous oxide are released. As a result, avoid placing it near the flame, and if you have to heat it, be sure to do it under the hood and observe safety tips.


First-aid measures:
- Skin Contact: Immediately flush skin with water.
- Inhalation: move the person to the fresh air.
- Eye Contact: Rinse eyes with water for at least 15-20 minutes.
- Ingestion: Do not induce vomiting. Get medical aid immediately. Call a poison control center.
Packing and Storage:
Keep the container tightly closed in a dry, cool, and well-ventilated place away from heat and sunlight.






Reviews
There are no reviews yet.